Novel MRI and PET markers of neuroinflammation in multiple sclerosis
Why is this important to me?
If you have read some of the other articles and book chapters on the causes of MS, you may recall that one trigger for MS is disruption of the blood-brain barrier that normally prevents immune cells circulating in the bloodstream from entering the brain and causing inflammation of brain tissues. This inflammation in turn contributes to the formation of lesions associated with MS. The term used for this process is "neuroinflammation." Monitoring of this harmful neuroinflammation is important for understanding the progress of the disease and for assessing an individual’s response to therapies. Assessment of neuroinflammation is typically done with a type of imaging called magnetic resonance imaging or MRI. As healthcare professionals understand more of the complex processes taking place in neuroinflammation, however, they increasingly find that conventional MRI does not always help them see these processes well. In order to provide a better view of the subtle processes of neuroinflammation, researchers and healthcare professionals are turning to new types of MRI as well as new contrast agents (called "tracers") to help them see what is happening in the individual's brain more clearly.
What is the objective of this study?
If we can see the process of neuroinflammation occurring earlier, we may be able to begin treatment earlier. We may also be better able to see when different disease-modifying therapies begin to work. The authors discuss different types of MRI as well as tracers that can improve our ability to understand what is taking place during neuroinflammation. They also discuss another imaging technology called positron emission tomography, usually abbreviated as PET.
First, let's look at conventional MRI:
- Conventional MRI detects acute neuroinflammation when used with an injectable tracer called gadolinium. Gadolinium-enhanced MRI can show active MS lesions that occur following disruption of the blood-brain barrier.
- Conventional MRI has limitations:
- Although gadolinium is generally considered safe to use in individuals who do not have kidney disease, some evidence suggests that accumulation of gadolinium may be toxic.
- In most individuals, the stages of neuroinflammation that conventional MRI enables us to view do not seem to match the stages of MS that the patient is experiencing. Again, we need to be able to see more detailed images.
- The various aspects of inflammation, such as different immune cells, different types of damage to the brain caused by inflammation, and different locations of inflammatory lesions, that cannot be seen with conventional MRI.
To overcome some of these limitations, newer MRI techniques and tracers are being developed to detect more changes in the brain. One new technique is called "susceptibility-weighted images" (SWI). The presence of iron and oxygen-rich blood can change the ability of MRI to create images of the brain. SWI can help to detect and measure:
- Myelin density. As you know, myelin is the fat-like substance that surrounds and protects nerve fibers. Immune cells crossing the blood-brain barrier attack and destroy myelin. If we are able to measure decreases in myelin density, we may be able to detect neuroinflammation earlier.
- Lesion central veins. Lesions appear to develop a central vein as they become more extensive. SWI can help us to monitor the development of central veins in lesions.
Another interesting technique is called "perfusion MRI," which measures changes in blood flow in the brain. Perfusion MRI could be used to:
- Determine how much leakage is occurring in an individual's blood-brain barrier and understand better how the amount of leakage is linked to the level of neuroinflammation taking place
- Understand better how the amount of blood and rate of blood flow in different areas of the brain are related to the stages of MS
- Inflammation in regions just below the surface of the brain (called leptomeningeal inflammation). These areas of inflammation occur in early and late MS and are distinct from the lesions detected with conventional MRI. These lesions are more common in progressive MS than relapsing-remitting MS.
Positron Emission Tomography (PET): MRI allows us to view tissues and structures in the brain. PET lets us see brain activity and function. PET works by using radioactive tracers that bind to specific molecules in the brain. PET may be able to help us understand more about how MS develops. One important piece of this puzzle is the role of cells called microglia. Unlike immune cells that cross the blood-brain barrier to enter the brain, microglia act as the brain's own resident immune cells. They inspect the complex cells and tissues of the brain to sweep away debris and damaged cells. Given the complexity of the brain, microglia are extremely sensitive to inflammation.
This is where things get fuzzy: we know that microglia and immune cells entering the brain appear to interact, but we do not know exactly how. We also know that these interactions are different depending on whether an individual has relapsing-remitting or primary progressive MS. Among the questions that PET may help to answer are:
- Are microglia or immune cells the primary cause of neuroinflammation?
- Does their interaction make neuroinflammation worse?
- Could microglia help to repair tissues damaged by neuroinflammation?
Several tracers are being developed for use in MRI and PET imaging that are expected to provide more precise and detailed information about inflammation in the brain. Therapeutic drugs can also be labeled with radioactive molecules and used to detect where in the brain these drugs act. Overall, the ability to visualize more detailed information about inflammation in your brain will provide new insight into disease progression, disability, your response to therapy, and how drugs work in your brain.
How did the authors study this issue?
The authors reviewed conventional and investigational MRI and PET methods and tracers for detecting inflammation in the brain of people with MS and related diseases.
| SHARE: | |||||
Original Article
Novel MRI and PET markers of neuroinflammation in multiple sclerosis
Marloes Hagens, Bart van Berckel, and Frederik Barkhof
Current Opinion in Neurology
ABSTRACT
Purpose of review: Gadolinium-enhancement depicts blood–brain barrier disruption associated with new inflammatory MRI lesions in multiple sclerosis (MS) and is widely used for diagnosis and therapeutic monitoring. However, earlier and more specific markers of inflammation are urgently needed.
Recent findings: Susceptibility-weighted images demonstrate the importance of the central vein in the formation of MS lesions. Perfusion weighted imaging techniques can show focal and diffuse low-grade inflammatory changes not visible on conventional MRI. Leptomeningeal enhancement could be part of the aetiology of subpial cortical MS lesions. Ultrasmall superparamagnetic particles of iron oxide can identify neuroinflammatory changes in addition to gadolinium enhancement and as such identify different types and phases of MS lesions. 18kD-translocator protein PET tracers identify activated microglia and an increase in TSPO uptake in both MS lesions and normal appearing brain tissue is related to disease severity and progression. A range of novel tracers for microglia activation is under development as well as radioligands that can label therapeutic drugs.
Summary: Novel MRI and PET techniques improve in-vivo visualization and quantification of the pleomorphic aspects of neuroinflammation, providing us with a unique insight in its pathogenesis, clinical relevance, and therapy responsiveness in MS.
INTRODUCTION
Multiple sclerosis (MS) is a complex immune-mediated disorder of the brain and spinal cord characterized by neuroinflammation, demyelination, gliosis, axonal degeneration, and neuronal loss. During the past decades imaging techniques such as magnetic resonance imaging (MRI) have contributed considerably to the improvement of clinical decision-making and the design of clinical trials in MS. Additionally, a range of novel tools using advanced MRI or PET technologies have been emerging from which we can obtain unique insights into pathogenesis of MS. The present article will summarize current developments in MRI and PET techniques to depict neuroinflammation in MS.

MRI
Conventional MRI techniques, such as T2 and T1-weighted sequences, are generally used in the diagnosis, follow-up, and therapeutic consideration in individual MS patients and clinical trials. Currently, the most commonly used MRI marker for acute inflammation is gadolinium-enhancing white matter lesions (WML), indicating disrupted blood–brain barrier (BBB) in these active MS lesions. Gadolinium-based contrast agents (GBCA) are generally considered safe in patients without severe renal insufficiency, but there is a potential risk for anaphylactoid-type reactions [1]. Moreover, recent studies suggest accumulation of (potentially toxic) gadolinium may occur in brain tissue in the absence of any significant renal dysfunction [2▪,3,4]. Apart from the potential side effects of GBCA, the correlation between conventional MRI markers for neuroinflammation and the clinical manifestations of MS in an individual patient is modest. Histopathological studies have shown that (inflammatory) brain pathology in MS is much more diverse and widespread than identified with conventional MRI techniques alone. Various developments regarding in-vivo MRI have advanced our understanding of the pleomorphic aspects of neuroinflammation in MS in recent years.
SUSCEPTIBILITY-WEIGHTED IMAGES
The magnetic property of brain tissue is largely determined by the presence of paramagnetic iron, oxygen levels of the blood in capillaries and veins and the amount of diamagnetic myelin and tissue water. Susceptibility-weighted images (SWI) uses the differences in magnetic susceptibility in brain tissue to identify MS-related tissue changes, such as iron deposition, myelin density, and venous capillary network density. To provide a quantitative measure for susceptibility, quantitative susceptibility mapping was developed. However, these techniques cannot disentangle the different and sometimes opposing factors that increase or decrease tissue susceptibility [5]. Considering the pathophysiology of neuroinflammatory lesions in MS, one of the most striking results from SWI studies is the identification of the central vein in lesions development. Such a central vein has been demonstrated in both periventricular and subcortical MS lesions and is significantly less frequent in non-MS WMLs [6–8]. Moreover, in a longitudinal 7 Tesla study by Dal-Bianco et al., an increase in volume of central veins of MS lesions was seen in the development of such a lesion [9▪]. This underlines the primal role of the central vein in lesion formation. In agreement with this, a recent retrospective 3 and 7 T MRI study by Absinta et al. demonstrated subtle signal changes on T2-weighted fluid-attenuated inversion recovery (T2-FLAIR) and T2* images up to 2 months before onset of focal gadolinium enhancing WML, that colocalize with the developing lesion's central vein [10▪]. Finally, SWI images can depict areas with phase shift without signal increase in other sequences, which may reflect areas of microglia activation (preactive lesions) [11].
PERFUSION-WEIGHTED IMAGING
Perfusion MRI is a collective term for three MR techniques that measure changes in cerebral haemodynamics. Dynamic contrast-enhanced (DCE) MRI measures the integrity of the BBB using GBCA as an in-vivo marker of low-grade neuroinflammation. Increased permeability of the BBB because of neuroinflammation enables extravasation of GBCA and the extravascular accumulation of such contrast agents increases the signal intensity in T1-weighted MRI images. By repeated acquisition of T1-weighted images during infusion of GBCA, DCE-MRI reveals tissue properties at a microvascular level, providing quantitative information on BBB integrity and leakage space as measures of (low-grade) neuroinflammation within and outside visible lesions [12▪▪,13].
Dynamic susceptibility contrast-enhanced (DSC) MRI measures cerebral blood volume and flow by monitoring the first pass of a bolus of GBCA by a series of T2*-weighted MRI images. Regional decreases in blood volume and flow have been reported in different stages of MS [14,15▪,16].
In contrast to the previous two methods, arterial spin labelling (ASL) is a noninvasive perfusion technique that uses magnetization of blood as an endogenous contrast agent [17▪,18,19]. In an ASL study using multiple delay times, an increase in bolus arrival time was demonstrated, indicating reduced perfusion. This is suggested to be caused by widespread arteriolar vasodilation associated with neuroinflammation [17▪].
In various recent studies both an increase and a decrease in cerebral perfusion have been linked to neuroinflammatory changes in MS and clinically isolated syndrome (CIS) [12▪▪,13,14,15▪,16,17▪,18,19]. Those contradictory results might imply changes in perfusion occur during inflammation. Standardization of acquisition techniques and postprocessing methods could help further implementation of perfusion-weighted MRI.
LEPTOMENINGEAL ENHANCEMENT
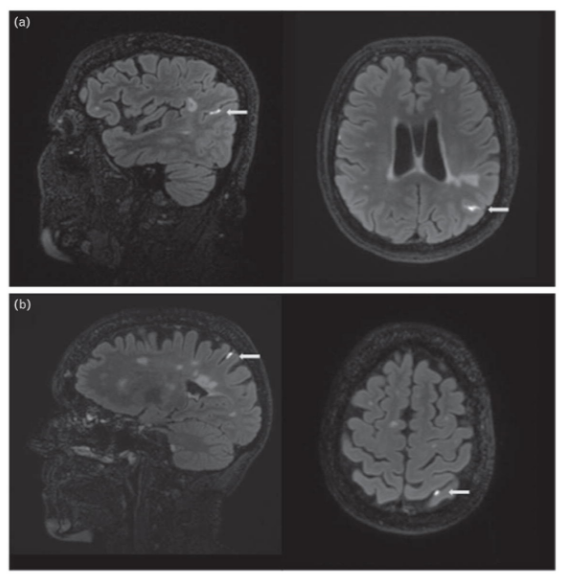
Besides the parenchymal GBCA enhancement used in the diagnoses and therapeutic decision making in MS, leptomeningeal enhancement has been described as part of the pathophysiology of MS (Fig. 1) [20]. Similar to the process in lesions, gadolinium enhancement of the leptomeninges is a result of vascular leakage and an indirect measure of leptomeningeal inflammation. Traditionally leptomeningeal enhancement is considered suggestive of diagnoses other than MS (e.g. sarcoid), but pathological studies have associated such leptomeningeal inflammation with subpial cortical demyelination and neurodegeneration in both early and late phases of MS [21,22]. In the last year, two MRI studies have aimed to quantify the prevalence and distribution of leptomeningeal inflammation in MS using 3 Tesla postcontrast T2-FLAIR MRI [23▪▪,24▪]. Absinta et al.[23▪▪] reported focal, usually perivascular, leptomeningeal enhancement in 74 of 299 MS patients and in one of 37 neurologically healthy controls. Strikingly, this prevalence was nearly twice as frequent in progressive MS patients (33%) compared to relapsing remitting MS (RRMS) (19%). In two cases that came to autopsy, pathologic evaluation confirmed the presence of perivascular leptomeningeal inflammation in three foci that enhanced in vivo, all in close relation to confluent cortical demyelination. On the contrary, Eisele et al. reported only one single case of focal leptomeningeal enhancement in a cohort of 122 MS patients of which only 15 had progressive MS. [24▪]. As a positive control group, five patients with stroke were included, all of whom showed leptomeningeal enhancement in a diffuse pattern. The conspicuous difference in prevalence between both studies could be attributed to the differences in patient population or in scanning techniques. This argues for further studies to determine the actual prevalence, natural history and clinical relevance of focal leptomeningeal enhancement in MS and its relation to cortical lesion formation.
ULTRASMALL SUPERPARAMAGNETIC PARTICLES OF IRON OXIDE
As mentioned before, gadolinium-enhancement indicates a defective BBB and is, therefore, an indirect measure of neuroinflammation in MS. In these lesions infiltrating blood-born monocytes play a crucial role, causing myelin breakdown and phagocytosing myelin debris. The phagocytic infiltration in the CNS can be directly detected in vivo with the use of ultrasmall superparamagnetic particles of iron oxide (USPIO) as an MRI contrast agent. Several hours after intravenous administration of USPIO these small nanoparticles are captured by phagocytic monocytes and accumulate in these cells [25]. Their iron oxide core distorts the magnetic field, causing a rapid decrease in T1 and T2-relaxation times of water molecules, resulting in an increased signal intensity on T1-weighted images and a decrease of signal on T2-weighted-gradient echo images [25,26]. Areas of altered intensity on delayed MRI scans presumably reflect the infiltration of USPIO-loaded macrophages in active MS lesions. USPIO enhancement can have one of three patterns: focal, ring-like or ‘return-to-isointensity’ (after having been hypointense on native T1-weighted images) [27]. Furthermore, global changes in the T1 signal in normal appearing brain tissue can demonstrate subtle generalized inflammatory activity without apparent BBB damage [28].
Clinical pilot trials have demonstrated that the use of both GBCA and USPIO in MS patients identifies more inflammatory lesions than gadolinium alone, ranging from a 4 to 244% increase [27,29,30]. The subgroup of lesions that enhanced with both contrast agents were characterized by a more severe evolution [27,30].
A recent study by Maarouf et al. in 18 CIS patients showed that USPIO enhancing lesions are already present at the onset of MS, but to a lesser extent than previously reported in RRMS patients [31▪]. The presence of such lesions was associated with more severe and persistent local tissue injury after 12 months of follow-up, but not with changes in Expanded Disability Status Scale (EDSS).
These limited number of studies show that USPIO allow for measurement of inflammation not possible with other MRI contrast agents, but the lack of available contrast agents limits further development.
PET
PET is a noninvasive molecular imaging technique to quantify biochemical and physiological processes in vivo. It measures the biodistribution of a radioligand or tracer, a radioactive isotope bound to biologically active molecule. Over the last decades, more and more new radioligands have been developed, creating a unique insight in the pathophysiology of neuroinflammation, neuronal dysfunction, demyelination, and remyelination. In this review we will focus on PET-tracers as biomarkers for neuroinflammation.
IMAGING-ACTIVATED MICROGLIA
The complex and highly dynamic processes of microglia activation is the central characteristic of neuroinflammation and a key element of neurodegeneration in MS [32,33]. Initially, in-vivo PET-studies investigating microglia activation used radioligands that bind to the 18kD-translocator protein (TSPO), formerly known as peripheral benzodiazepine receptor. The first successful and now most frequently used of the TSPO-radioligands is [11C]PK11195 [34]. [11C]PK11195 uptake is increased in focal T2 MRI lesions, colocalizes with gadolinium-enhancing T1 lesions in RRMS and is increased in normal appearing white and grey matter (NAWM and normal appearing grey matter) in MS patients compared with healthy controls [35–37]. Overall there is significant positive correlation with [11C]PK11195 uptake and disease duration, disability score and brain atrophy [35–38]. However, [11C]PK11195 has several disadvantages, including limited brain entrance, poor signal-to-noise ratio and labelling with the impractically rapid decaying isotope [11C] (t1/2 = 20.33 min) [39]. This led to the development of a wide range of second generation TSPO radioligands; see Table 1 for an overview of TSPO PET studies in MS patients. The main disadvantage of these new tracers is single nucleotide polymorphisms (SNPs) in the TSPO gene (rs697) that induce a variation in binding activity for these second generation TSPO tracers [50]. Differences in affinity between the high-affinity binding type and the low-affinity binding type varies between the different TSPO tracers from a four-fold to as high as a 50-fold decrease in binding [50].
Apart from the use of different tracers and genetic polymorphism, comparison of TSPO PET studies can be challenging because of differences in quantification methods of specific binding, as well as differences in patient population, such as subtypes of MS, disease duration, disease severity, and age. Recent studies have illustrated the relevance of imaging these different subgroups in further understanding the pathophysiology of the different disease stages in MS.
Giannetti et al. recruited 18 CIS patients for a [11C]PK11195 study [43▪▪]. They demonstrated a global increase in [11C]PK11195 uptake in NAWM and deep grey matter compared with healthy controls and a further significant increase in those CIS patients with T2 MRI lesions. Furthermore, higher levels of uptake were correlated with a development of MS at 2-year interval. This suggests that diffuse inflammatory changes in the NAWM in early phases of the disease predispose for WML development and subsequently conversion to MS. This would be in line with previous ex-vivo immune–histochemical studies, showing areas of activated microglia without apparent loss of myelin at risk of developing into acute inflammatory lesions [35,51,52].
Rissanen et al. used the same TSPO-tracer to image not early but chronic disease in the secondary progressive MS (SPMS) patients [41▪]. First of all they showed an increase in [11C]PK11195 uptake in 57% of black holes, which has recently been related to a higher degree of clinical disability by Giannetti et al.[42▪]. Moreover, there was a significant increase in tracer uptake in the global NAWM and in the thalamus in SPMS patients compared with healthy controls. This is in agreement with the pathology findings of a more chronic and diffuse low-grade inflammation behind the BBB in the progressive phase of the disease [53].
In the RRMS, there seems to be an association between disease activity and global microglia activity measured with TSPO PET. On one hand, Park et al. found no increase in [11C]PBR28 uptake in the NAWM of four clinically and radiologically stable RRMS patients [45▪]. However, Colasanti et al. described a positive relation between [18F]PBR111 uptake and disease severity scores in 11 RRMS patients [46▪].
Overall, increase in TSPO uptake appears to be related to disease severity and progression.
NOVEL PET TRACERS FOR ACTIVATED MICROGLIA
Besides TSPO radioligands, new PET tracers with different binding targets on microglia are under development.
In MS the purinergic, ATP binding, receptor P2X7 (P2X7R) is upregulated on microglia. Binding of ATP to the receptor activates the microglia, which leads to proliferation and release of the proinflammatory cytokine and subsequently microglia recruitment [54]. Recently, ATP-dependent release of tumour necrosis factor α was demonstrated to have a neuroprotective effect in MS [55]. Moreover, a rare SNP rs28360457 in the P2X7R gene has been described to protect against the risk of developing MS [56▪]. All in all, P2X7R is an interesting target for PET tracers and potentially also for drug development. A first P2X7R PET-tracer [11C]A-740003 is under evaluation in animal models of neuroinflammation by Janssen et al.[57]. Gao et al. developed a second radioligand [11C]GSK1482160 which has yet to be evaluated in vivo[58].
Secondly, the adenosine A2A receptor (A2AR) has been proposed as a new in-vivo target for imaging neuroinflammation. In-vitro studies show inflammatory stimuli lead to upregulation of A2AR on microglial cells and subsequently alter their morphology and behaviour [59]. This possibly provides an endogenous pathway to limit neuroinflammation and alter the pathophysiology process of neurodegeneration [60]. Rissanen et al. showed an increased uptake of the A2AR PET-tracer [11C]TMSX in NAWM of MS patients compared with healthy controls, which was associated with higher EDSS and increased brain tissue loss [61,62]. On the downside, [11C]TMSX suffers from low-binding potentials and high-nonspecific binding. Therefore, new A2AR tracers are currently under investigation.
LABELLING DRUGS THAT TARGET NEUROINFLAMMATION
Furthermore, PET tracers have been developed to analyse the organ and tissue distribution of drugs currently used in or under development for treatment of MS. As these drugs usually have a long pharmacokinetic half-life the long-lived positron emitters, [124I] (t1/2 = 4.2 days) and [89Zr] (t1/2 = 3.3 days) are more commonly used.
To quantify the brain penetration of siponimod (BAF312), a selective sphingosine-1-phosphate receptor (S1PR) agonist in phase 3 development for treatment of SPMS, Briard et al. developed compound MS565 [63,64]. By slightly modifying the structure of BAF312 this molecule can be labelled to [124I] for PET imaging. Despite the additional iodine atom the biochemical properties and pharmacokinetics of [124I]MS565 remain very close to the original compound. In a similar fashion they previously developed [124I]BZM055 as a surrogate tracer to study the biodistribution of fingolimod (FTY720), a S1PR agonist approved for the treatment of RRMS [65]. Further validation of these radioligands is necessary.
In contrast to radioligands based on MS-specific small molecules, radioactive labelling of mAbs has already been validated in oncology [66]. Similar studies in MS patients can give us important insights in the mechanisms responsible for the therapeutic effect of mAbs in MS. At ECTRIMS 2015 a PET study with [89Zr]-labelled rituximab, a chimeric mAb against the CD20 molecule on mature B lymphocytes used in the treatment of RRMS, was presented [67]. There was no evidence of cerebral penetration for rituximab in this pilot study with two active RRMS patients, suggesting the (early) therapeutic effects of this drug are independent of CNS penetration. Further larger studies with labelled mAbs are highly relevant, especially in light of the recently reported phase III result of the ocrelizumab trials in MS [68].
CONCLUSION
Current advances in MRI and PET techniques have expanded the armamentarium for in vivo visualization and quantification of the pleomorphic aspects of neuroinflammation in MS, providing us with a unique insight in its pathogenesis, clinical relevance and therapy responsiveness not possible with conventional MRI techniques. Further work should aim at validating these novel imaging methods and ideally implementing them in the clinical practice and drug development.
ACKNOWLEDGEMENTS
The authors wish to thank Dr J. Killestein, Prof. Dr A.D. Windhorst, Prof. Dr A.A. Lammertsma, Dr A.M.W. van Dam and Prof. Dr H.E. de Vries for their help on various projects.
Financial support and sponsorship
The work was supported by MS Center Amsterdam and Neuroscience Campus Amsterdam.
Conflicts of interest
M.H. and B.v.B. have no conflicts of interest.
F.B. serves on the editorial boards of Brain, Neurology, Neuroradiology, Multiple Sclerosis Journal and Radiology, and serves as a consultant for Bayer-Schering Pharma, Sanofi-Aventis, Genzyme, Biogen-Idec, Teva, Novartis, Roche, Synthon BV, and Jansen Research.
REFERENCES AND RECOMMENDED READING
Papers of particular interest, published within the annual period of review, have been highlighted as:
▪ of special interest
▪▪ of outstanding interest
REFERENCES
1. Khawaja AZ, Cassidy DB, Al Shakarchi J, et al. Revisiting the risks of MRI with gadolinium based contrast agents – review of literature and guidelines. Insights Imaging 2015; 6:553–558.
2▪. Kanda T, Ishii K, Kawaguchi H, et al. Hyperintensity in dentate nucleus and globus pallidus on unenhanced MR images kanda et al. Radiology 2014; 270:834–841.
This is the first study to describe a cumulative effect of previous GBCA administrations on the signal intensity in the dentate nucleus and globus pallidus of unenhanced T1-weighted MR images.
3. Errante Y, Cirimele V, Mallio CA, et al. Progressive increase of T1 signal intensity of the dentate nucleus on unenhanced magnetic resonance images is associated with cumulative doses of intravenously administered gadodiamide in patients with normal renal function, suggesting dechelation. Invest Radiol 2014; 49:685–690.
4. McDonald RJ, McDonald JS, Kallmes DF, et al. Intracranial gadolinium deposition after contrast-enhanced MR imaging. Radiology 2015; 275:772–782.
5. Liu C, Li W, Tong Ka, et al. Susceptibility-weighted imaging and quantitative susceptibility mapping in the brain. J Magn Reson Imaging 2015; 42:23–41.
6. Kau T, Taschwer M, Deutschmann H, et al. The ‘Central Vein Sign’: is there a place for susceptibility weighted imaging in possible multiple sclerosis? Eur Radiol 2013; 23:1956–1962.
7. Maggi P, Mazzoni LN, Moretti M, et al. SWI enhances vein detection using gadolinium in multiple sclerosis. Acta Radiol 2015; 4:1–4.
8. Lane JI, Bolster B, Campeau NG, et al. Characterization of multiple sclerosis plaques using susceptibility-weighted imaging at 1.5 T: can perivenular localization improve specificity of imaging criteria? J Comput Assist Tomogr 2015; 39:317–320.
9▪. Dal-Bianco A, Hametner S, Grabner G, et al. Veins in plaques of multiple sclerosis patients – a longitudinal Magnetic Resonance Imaging Study at 7 Tesla. Eur Radiol 2015; 25:2913–2920.
This 7 T longitudinal study using SWI shows the volume of intralesional veins is significantly increased during the development of MS lesions, underlining the primal role of the central vein in lesion formation.
10▪. Absinta M, Nair G, Sati P, et al. Direct MRI detection of impending plaque development in multiple sclerosis. Neurol Neuroimmunol Neuorinflamm 2015; 2:e145.
Subtle signal changes on T2-FLAIR and T2* images can be seen up to 2 months before onset of focal gadolinium-enhancing WML.
11. Kakeda S, Futatsuya K, Ide S, et al. Improved detection of cortical gray matter involvement in multiple sclerosis with quantitative susceptibility mapping. Acad Radiol 2015; 22:1427–1432.
12▪▪. Cramer SP, Simonsen H, Frederiksen JL, et al. Abnormal blood-brain barrier permeability in normal appearing white matter in multiple sclerosis investigated by MRI. NeuroImage Clin 2014; 4:182–189.
Permeability of the BBB in periventricular NAWM measured by DCE-MRI, can significantly improve prediction of conversion to MS in optic neuritis patients, compared with T2 lesion count alone.
13. Cramer SP, Modvig S, Simonsen HJ, et al. Permeability of the blood – brain barrier predicts conversion from optic neuritis to multiple sclerosis. Brain 2015; 138:2571–2583.
14. Sowa P, Bjørnerud A, Nygaard GO, et al. Reduced perfusion in white matter lesions in multiple sclerosis. Eur J Radiol 2015; 84:2605–2612.
15▪. Bester M, Forkert ND, Stellmann JP, et al. Increased perfusion in normal appearing white matter in high inflammatory multiple sclerosis patients. PLoS One 2015; 10:e0119356.
DSC-MRI is sensitive to diffuse microvascular pathology in NAWM and this differentiates between high and low-inflammatory patients.
16. Papadaki EZ, Simos PG, Panou T, et al. Hemodynamic evidence linking cognitive deficits in clinically isolated syndrome to regional brain inflammation. Eur J Neurol Neurol 2014; 21:499–505.
17▪. Paling D, Thade Petersen E, Tozer DJ, et al. Cerebral arterial bolus arrival time is prolonged in multiple sclerosis and associated with disability. J Cereb Bood Flow Metab 2014; 34:34–42.
This ASL MRI study shows altered cerebral aterial haemodynamics in NAWM and deep grey matter is related to clinical disability scores, possibly resulting from widespread inflammatory changes.
18. Narayana Pa, Zhou Y, Hasan KM, et al. Hypoperfusion and T1-hypointense lesions in white matter in multiple sclerosis. Mult Scler J 2014; 20:365–373.
19. Debernard L, Melzer TR, Van Stockum S, et al. Reduced grey matter perfusion without volume loss in early relapsing-remitting multiple sclerosis. J Neurol Neurosurg Psychiatry 2014; 85:544–551.
20. Guseo A, Jellinger K. The significance of perivascular infiltrations in multiple sclerosis. J Neurol 1975; 211:51–60.
21. Lucchinetti CF, Popescu BFG, Bunyan RF, et al. Inflammatory cortical demyelination in early multiple sclerosis. N Engl J Med 2011; 365:2188–2197.
22. Choi SR, Howell OW, Carassiti D, et al. Meningeal inflammation plays a role in the pathology of primary progressive multiple sclerosis. Brain 2012; 135:2925–2937.
23▪▪. Absinta M, Vuolo L, Rao A, et al. Gadolinium-based MRI characterization of leptomeningeal inflammation in multiple sclerosis. Neurology 2015; 85:18–28.
This study demonstrates a rather high prevalence of leptomeningeal enhancement in especially progressive MS patients. The association with cortical MS lesions is highly relevant in understanding the pathophysiology of such lesions.
24▪. Eisele P, Griebe M, Szabo K, et al. Investigation of leptomeningeal enhancement in MS: a postcontrast FLAIR MRI study. Neurology 2015; 84:770–775.
Reporting a very low prevalence of leptomeningeal enhancement in MS in contrary to the previous reference, this article creates an interesting discussion on the importance of leptomeningeal enhancement in the pathophysiology of MS.
25. Dousset V, Ballarino L, Delalande C, et al. Comparison of ultrasmall particles of iron oxide (USPIO)-enhanced T2-weighted, conventional T2-weighted, and gadolinium-enhanced T1-weighted MR images in rats with experimental autoimmune encephalomyelitis. Am J Neuroradiol 1999; 20:223–227.
26. Corot C, Robert P, Idée JM, et al. Recent advances in iron oxide nanocrystal technology for medical imaging. Adv Drug Deliv Rev 2006; 58:1471–1504.
27. Vellinga MM, Oude Engberink RD, Seewann A, et al. Pluriformity of inflammation in multiple sclerosis shown by ultra-small iron oxide particle enhancement. Brain 2008; 131:800–807.
28. Vellinga MM, Vrenken H, Hulst HE, et al. Use of ultrasmall superparamagnetic particles of iron oxide (USPIO)-enhanced MRI to demonstrate diffuse inflammation in the normal-appearing white matter (NAWM) of multiple sclerosis (MS) patients: an exploratory study. J Magn Reson Imaging 2009; 29:774–779.
29. Dousset V, Brochet B, Deloire MS, et al. MR imaging of relapsing multiple sclerosis patients using ultra-small-particle iron oxide and compared with gadolinium. Am J Neuroradiol Neuroradiol 2006; 27:1000–1005.
30. Tourdias T, Stéphanie Roggerone M, Massimo Filippi M, et al. Assessment of disease activity in multiple sclerosis phenotypes with combined gadolinium- and superparamagnetic iron oxide-enhanced MR imaging. Radiology 2012; 264:225–233.
31▪. Maarouf A, Ferre JC, Zaaraoui W, et al. Ultra-small superparamagnetic iron oxide enhancement is associated with higher loss of brain tissue structure in clinically isolated syndrome. Mult Scler J 2015; [Epub ahead of print].
As the first USPIO study published in a number of years, this demonstrates macrophage infiltration early on in the disease and its relation to the severity of neuroinflammatory changes in the brain.
32. Hemmer B, Kerschensteiner M, Korn T. Role of the innate and adaptive immune responses in the course of multiple sclerosis. Lancet Neurol 2015; 14:406–419.
33. Mahad DH, Trapp BD, Lassmann H. Pathological mechanisms in progressive multiple sclerosis. Lancet Neurol 2015; 14:183–193.
34. Vowinckel E, Reutens D, Becher B, et al. PK11195 binding to the peripheral benzodiazepine receptor as a marker of microglia activation in multiple sclerosis and experimental autoimmune encephalomyelitis. J Neurosci Res 1997; 50:345–353.
35. Banati RB, Newcombe J, Gunn RN, et al. The peripheral benzodiazepine binding site in the brain in multiple sclerosis: quantitative in vivo imaging of microglia as a measure of disease activity. Brain 2000; 123:2321–2337.
36. Debruyne JC, Versijpt J, Laere van KJ, et al. PET visualization of microglia in multiple sclerosis patients using [11C]PK11195. Eur J Neurol 2003; 10:257–264.
37. Politis M, Giannetti P, Su P, et al. Increased PK11195 PET binding in the cortex of patients with MS correlates with disability. Neurology 2012; 79:523–530.
38. Versijpt J, Debruyne JC, Laere van KJ, et al. Microglial imaging with positron emission tomography and atrophy measurements with magnetic resonance imaging in multiple sclerosis: a correlative study. Mult Scler J 2005; 11:127–134.
39. Kreisl WC, Fujita M, Fujimura Y, et al. Comparison of [11C]-(R)-PK 11195 and [11C]PBR28, two radioligands for translocator protein (18 kDa) in human and monkey: implications for positron emission tomographic imaging of this inflammation biomarker. Neuroimage 2010; 49:2924–2932.
40. Ratchford JN, Endres CJ, Hammoud DD, et al. Decreased microglial activation in MS patients treated with glatiramer acetate. J Neurol 2012; 259:1199–1205.
41▪. Rissanen E, Tuisku J, Rokka J, et al. In vivo detection of diffuse inflammation in secondary progressive multiple sclerosis using PET imaging and the radioligand 11 C-PK11195. J Nucl Med 2014; 55:939–944.
This in-vivo TSPO PET study supports earlier neuropathological studies identifying the pivotal role of microglia in low-grade inflammation behind intact BBB in the SPMS.
42▪. Giannetti P, Politis M, Su P, et al. Microglia activation in multiple sclerosis black holes predicts outcome in progressive patients: an in vivo [(11)C](R)-PK11195-PET pilot study. Neurobiol Dis 2014; 65:203–210.
A heterogenous binding pattern of [11C]PK11195 in black holes related to the severity of the disease course in progressive MS, indicates clinically relevant inflammatory activity in these ‘holes’.
43▪▪. Giannetti P, Politis M, Su P, et al. Increased PK11195-PET binding in normal-appearing white matter in clinically isolated syndrome. Brain 2015; 138:110–119.
This is a first TSPO PET study in CIS patients. It identifies diffuse inflammatory changes in the NAWM that predispose for WML development and subsequently conversion to MS.
44. Oh U, Fujita M, Ikonomidou VN, et al. Translocator protein PET imaging for glial activation in multiple sclerosis. J Neuroimmune Pharmacol 2011; 6:354–361.
45▪. Park E, Gallezot J-D, Delgadillo A, et al. 11C-PBR28 imaging in multiple sclerosis patients and healthy controls: test-retest reproducibility and focal visualization of active white matter areas. Eur J Nucl Med Mol Imaging 2015; 42:1081–1092.
The test–retest study demonstrates the reliability of the second-generation TSPO radioligand [11C]PBR28 in MS.
46▪. Colasanti A, Guo Q, Muhlert N, et al. In vivo assessment of brain white matter inflammation in multiple sclerosis with [18F]-PBR111 PET. J Nucl Med 2014; 55:1112–1118.
This is a first study using the second-generation TSPO tracer [18F]PBR111 in MS patients. It shows a positive correlation between [18F]PBR111 uptake and MS severity scores in RRMS.
47. Vas Á, Shchukin Y, Karrenbauer VD, et al. Functional neuroimaging in multiple sclerosis with radiolabelled glia markers: Preliminary comparative PET studies with [11C]vinpocetine and [11C]PK11195 in patients. J Neurol Sci 2008; 264:9–17.
48. Takano A, Piehl F, Hillert J, et al. In vivo TSPO imaging in patients with multiple sclerosis: a brain PET study with [18F]FEDAA1106. Eur J Nucl Med Mol Imaging Res 2013; 3:30.
49. Trigg W, Buckley C, Heurling K, et al. Evaluation of [18F]GE-180 PET imaging in relapsing-remitting multiple sclerosis patients: a first-in-human study [abstract]. ECTRIMS Online Libr 2015; 115715.
50. Owen DRJ, Gunn RN, Rabiner EA, et al. Mixed-affinity binding in humans with 18-kDa translocator protein ligands. J Nucl Med 2011; 52:24–32.
51. Marik C, Felts PA, Bauer J, et al. Lesion genesis in a subset of patients with multiple sclerosis: a role for innate immunity? Brain 2007; 130:2800–2815.
52. Van Horssen J, Singh S, van der Pol S, et al. Clusters of activated microglia in normal-appearing white matter show signs of innate immune activation. J Neuroinflammation 2012; 9:156.
53. Allen IV, McQuaid S, Mirakhur M, et al. Pathological abnormalities in the normal-appearing white matter in multiple sclerosis. Neurol Sci 2001; 22:141–144.
54. Monif M, Burnstock G, Williams DA. Microglia: proliferation and activation driven by the P2X7 receptor. Int J Biochem Cell Biol 2010; 42:1753–1756.
55. Masuch A, Shieh C-H, van Rooijen N, et al. Mechanism of microglia neuroprotection: involvement of P2X7, TNFα, and valproic acid. Glia 2016; 64:76–89.
56▪. Gu BJ, Field J, Dutertre S, et al. A rare P2X7 variant Arg307Gln with absent pore formation function protects against neuroinflammation in multiple sclerosis. Hum Mol Genet 2015; 24:5644–5654.
A rare loss of function SNP in the P2X7 gene has a 1.8-fold protective effect on the risk of developing MS in a large Australian and a large European cohort.
57. Janssen B, Vugts DJ, Funke U, et al. Synthesis and initial preclinical evaluation of the P2X7 receptor antagonist [11C]A-740003 as a novel tracer of neuroinflammation. J Label Compd Radiopharm 2014; 57:509–516.
58. Gao M, Wang M, Green MA, et al. Synthesis of [11C]GSK1482160 as a new PET agent for targeting P2X7 receptor. Bioorg Med Chem Lett 2015; 25:1965–1970.
59. Orr AG, Orr AL, Li X-J, et al. Adenosine A2A receptor mediates microglial process retraction. Nat Neurosci 2009; 12:872–878.
60. Santiago AR, Baptista FI, Santos PF, et al. Role of microglia adenosine A(2A) receptors in retinal and brain neurodegenerative diseases. Mediators Inflamm 2014; 2014:465694.
61. Rissanen E, Virta JR, Paavilainen T, et al. Adenosine A2A receptors in secondary progressive multiple sclerosis: a [(11)C]TMSX brain PET study. J Cereb Blood Flow Metab 2013; 33:1394–1401.
62. Rissanen E, Tuisku J, Luoto P, et al. Automated reference region extraction and population-based input function for brain [(11)C]TMSX PET image analyses. J Cereb Blood Flow Metab 2015; 35:157–165.
63. Briard E, Rudolph B, Desrayaud S, et al. MS565: A SPECT tracer for evaluating the brain penetration of BAF312 (siponimod). ChemMedChem 2015; 10:1008–1018.
64. Tavares A, Barret O, Alagille D, et al. Brain distribution of MS565, an imaging analogue of siponimod (BAF312), in non-human primates. Neurology 2014; 82:P1.168.
65. Briard E, Orain D, Beerli C, et al. BZM055, an iodinated radiotracer candidate for PET and SPECT imaging of myelin and FTY720 brain distribution. ChemMedChem 2011; 6:667–677.
66. Wright BD, Lapi SE. Designing the magic bullet? The advancement of immuno-PET into clinical use. J Nucl Med 2013; 54:1171–1174.
67. Hagens MHJ, Killestein J, Dongen GAMS van, et al. Imaging of rituximab-zirconium-89 uptake with PET scans in active relapsing-remitting MS patients [abstract]. ECTRIMS Online Libr 2015; 116006.
68. Montalban X, Hemmer B, Rammohan K, et al. Efficacy and safety of ocrelizumab in primary progressive multiple sclerosis – results of the placebo-controlled, double-blind, phase III ORATORIO Study [abstract]. ECTRIMS Online Libr 2015; 116701.

