Predictors of Fatigue Impact in Persons With Long-Standing Multiple Sclerosis
Why is this important to me?
All of us experience fatigue, but MS-related fatigue is a uniquely complex occurrence that many individuals with MS experience. The Multiple Sclerosis Council for Clinical Practice Guidelines defines MS-related fatigue as ‘‘a subjective lack of physical and/or mental energy that is perceived by the individual or caregiver to interfere with usual and desired activities.’’
Despite being one of the most common and irritating symptoms of MS, we do not understand MS-related fatigue well, and most therapies are not especially effective. MS-related fatigue is complicated because it seems to be caused by disorders in the central nervous system as well as factors such as depression and loss of physical function.
Although it is easy to find reports on the experience of fatigue among individuals with MS, researchers have not looked at fatigue among individuals who have lived with MS for some time. If we can better understand what conditions or factors seem to predict or contribute to fatigue in individuals who have been living for a long time with MS, we can begin to study how to reduce the impact of MS-related fatigue.
What is the objective of this study?
In this study, the authors explored the relationships between people with longstanding MS and these conditions:
- Age — does advanced age make one more likely to develop fatigue?
- Physical limitations — does loss of physical capabilities contribute to fatigue?
- Symptoms of depression — to what extent does depression predict fatigue?
- Personal resources — does lack of care make one likely to become fatigued?
- Health-promoting behavior — are individuals who live healthily able to avoid symptoms of fatigue?
- Barriers to health promotion — conversely, do barriers to healthy activities and behavior predispose individuals to fatigue?
Their findings are interesting:
- There appears to be no relationship between age and length of life with MS. This suggests that fatigue may be experienced throughout the disease course irrespective of age or duration of MS.
- Functional limitation seems to be the strongest predictor of physical and psychosocial fatigue, while symptoms of depression are the strongest predictor of total and cognitive fatigue in this sample.
- The weakest predictor of total, physical, and psychosocial fatigue is barriers to health-promoting activities. In other words, it pays to “live wise!”
- Physical impairment was the weakest predictor of cognitive fatigue.
It makes sense that individuals who experience problems with their functional or physical capabilities would be more likely to develop physical and psychosocial fatigue. Similarly, we can understand that depression and symptoms of depression could predict cognitive as well as total fatigue. The relationship between fatigue and depression is quite complex and has numerous symptoms in common (sleep disturbances and low motivation, to name two), although the exact cause remains yet to be explained.
Overall, the researchers identified functional impairment, depression, and barriers to health-promoting activities as the factors associated with the likelihood of fatigue in individuals with long-standing MS. Additional research is needed to explore if interventions to enhance management of depression and reducing barriers to health-promoting activities would result in lower fatigue among persons with long-standing MS.
These results uphold the importance of communicating changes in your mood and functional status as well as experience with fatigue at each opportunity that you meet with your healthcare provider.
How did the authors study this issue?
This study was conducted with a series of surveys that were answered by 331 people who had been living with MS for more than 17 years and who resided in the southwestern US.
Participants answered surveys designed to assess:
- How fatigue has impacted the participant’s life over the past 4 weeks. This self-reported survey (called the Modified Fatigue Impact Scale or MFIS) assesses the effects of fatigue on physical, cognitive, psychosocial, and total function.
- Functional limitation due to MS
- Depression
- Healthy behavior, and barriers to healthy activities
- Personal resources such as social support
The participants were 87% women, with an average age of 63 years and an average time living with MS of 27 years. After collecting the responses to the surveys, the authors performed statistical analyses to determine factors that were predictors of the impact of fatigue on function and daily activities.
| SHARE: | |||||
Original Article
Predictors of Fatigue Impact in Persons With Long-Standing Multiple Sclerosis
Journal of Neuroscience Nursing
Janet D. Morrison, Alexa K. Stuifbergen
Background: Fatigue is a common experience among persons diagnosed with multiple sclerosis (MS). Fatigue negatively influences quality of life, interferes with activities of daily living, and impairs the ability to maintain gainful employment. Mechanisms underlying the pathophysiologic determinants of fatigue in MS are poorly understood, and effective treatments to manage fatigue present a challenge. Although the use of pharmacologic therapies is recommended to treat symptomatic fatigue, undertreatment of fatigue is common. Better long-term management and symptomatic relief may be provided by the use of nonpharmacologic treatments such as increased physical activity, energy conservation, and cognitive behavioral therapy.
Methods: The purpose of this study was to explore the physical, cognitive, and psychosocial dimensions of fatigue impact among persons with long-standing MSVdefined here as having been diagnosed with MS for 17 years or more. A sample of 331 participants with MS completed surveys measuring fatigue impact, MS-related functional limitation, depressive symptoms, barriers to health promotion, personal resources, and health promoting behaviors.
Results: MS-related functional limitation and depressive symptoms were the strongest predictors of fatigue impact. MS-related functional limitation explained the greatest amount of variance in models predicting physical and psychosocial fatigue impact, whereas depressive symptoms explained the most variance in models predicting total and cognitive fatigue impact. Barriers to health promotion explained the least amount of variance among the models. Personal resources and health promoting behaviors were not significant predictors of fatigue impact in this study.
Conclusion: Interventions aimed at reducing MS-related functional limitations, depressive symptoms, and barriers to health promotion may have beneficial influences on fatigue impact, whereas actions designed to promote personal resource adequacy as well as engagement in health promoting behaviors may not translate into improvements in fatigue impact in persons with MS.
Background
Multiple sclerosis (MS) is the most common neurological disease of young adults affecting 2.3 million persons worldwide and approximately 140 per 100,000 in North America (Multiple Sclerosis International Federation, 2013). Fatigue related to MS is the most frequently reported and arguably among the most disabling symptoms of the disease. Most persons with MS experience significant fatigue on a daily basis (Fisk, Pontefract, Ritvo, Archibald, & Murray, 1994). In a nationally representative sample of over 4,000 individuals with MS, more respondents cited fatigue (83%) as a current symptom than difficulties with ambulation (67%), spasticity (63%), or cognitive problems (56%; Minden et al., 2006). Fatigue negatively impacts social, emotional, cognitive, and physical functioning in those with MS and is related to lower health-related quality of life and unemployment (Krupp, 2003). Despite MS-related fatigue being so prevalent, its pathophysiology is not well understood, and therapeutic regimens lack robust effects (Kluger, Krupp, & Enoka, 2013).
Whereas fatigue is a ubiquitous human experience, MS-related fatigue is a uniquely complex, multifactorial phenomenon that, for the purposes of this article, is defined as ‘‘a subjective lack of physical and/or mental energy that is perceived by the individual or caregiver to interfere with usual and desired activities’’ (Multiple Sclerosis Council for Clinical Practice Guidelines, 1998). Appraisal of fatigue in MS presents unique challenges because fatigue is hypothesized to be derived from primary sources directly related to MS pathology (e.g., lesions in the central nervous system, hypothalamus–pituitary–adrenal axis dysfunction) as well as secondary sources related to coexisting features of MS such as depression and physical deconditioning (Induruwa, Constantinescu, & Gran, 2012; Krupp, Serafin, & Christodoulou, 2010). Strategies to assess fatigue include both self-report and performance-based measures. Self-report measures such as the Modified Fatigue Impact Scale (MFIS; Fisk, Ritvo, et al., 1994) aim to capture perceptions of fatigue impact on a variety of daily activities, whereas others, like the Fatigue Severity Scale (Krupp, LaRocca, Muir-Nash, & Steinberg, 1989), depict fatigue intensity (Krupp & Christodoulou, 2001).
Although much has been reported on the experience of fatigue in persons with MS, scant attention has been focused on individuals living with the disease for extended periods. The current study examined individuals who have all been diagnosed with MS for over 17 years. The study sample has a mean disease duration of 26.5 ± 6.5 years. The authors define this phenomenon as long-standing MS. Little is known of factors that covary with and, therefore, may be predictive of fatigue among persons with long-standing MS. A better understanding of factors predictive of fatigue in persons with long-standing MS can inform intervention studies designed to reduce the impact of fatigue in persons living with MS.
Purpose
Dimensions of fatigue impact- total, physical, cognitive, and psychosocial- among persons with longstanding MS are the focus of this article. The purpose of this study was to investigate relationships among self-reported ratings of fatigue impact, age, MS duration, MS-related functional limitations, personal resources, depressive symptoms, health promoting behaviors, and barriers to health promotion. Two research questions were addressed using Pearson’s correlations and multiple regression analyses:
- What relationships exist among measures of fatigue impact, age, MS duration, MS-related functional limitations, personal resources, depressive symptoms, health promoting behaviors, and barriers to health promotion in this sample of persons with long-standing MS?
- What amount of variance in fatigue impact, measured by the MFIS, can be explained by significantly related variables among persons with long-standing MS?
Methods
Participants and Procedures
For the current study, data were derived from the 17th year of a longitudinal survey study of persons with MS residing in the community. In 1999, after approval from the university’s institutional review board, respondents from a 1991 cross-sectional study who expressed interest in continuing to participate in a longitudinal descriptive study were consented. The original study was recruited through selected chapters of the National MS Society and announcements in several rural southwestern newspapers (Stuifbergen & Roberts, 1997). Participants in the longitudinal study are mailed a survey each year through the U.S. Postal Service that contains selected self-report measures of health promoting behaviors, MS-related functional limitations, barriers to health promotion, personal resources, depressive symptoms, and quality of life. A measure of fatigue impact was included in the 2013 survey. The current analysis examined responses to selected variables for the 331 individuals responding to the 2013 survey.
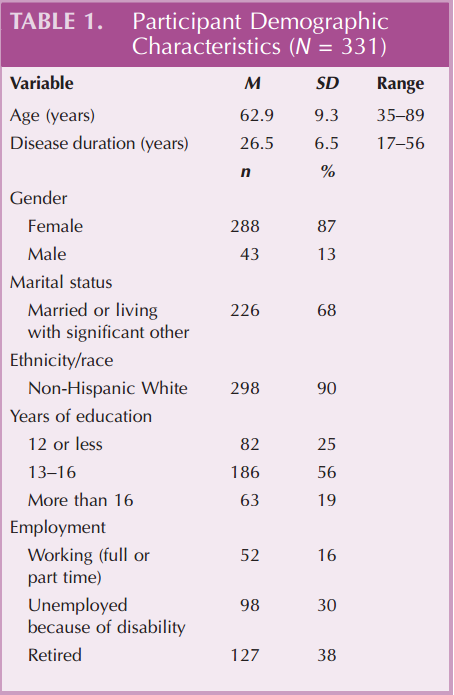
The sample for this study is well educated (75% have 12+ years of formal education), married or living with a significant other (68%), and mostly female (87%). These demographic characteristics are common among MS studies. Participants ranged in age from 35 to 89 years with an average of 62.9 T 9.3 years. Average MS duration was 26.5 T 6.5 years. Responses to questions about employment indicate that 30% were unemployed because of disability and 38% were retired, whereas 16% were employed either full or part-time (see Table 1).
Measures
Demographic Data
Background demographic characteristics were collected in the third and seventh years of the longitudinal study regarding age, gender, year diagnosed with MS, race, ethnicity, and education. Employment and marital status data are reported from the current 2013 survey.
Fatigue
The MFIS is a 21-item form of the Fatigue Impact Scale (Fisk, Ritvo, et al., 1994) derived from interviews with 105 patients with MS by the Consortium of Multiple Sclerosis Centers Health Services Research Subcommittee (1997). The MFIS assesses the impact of fatigue on the lives of persons with MS over the previous 4 weeks yielding a total (21-item) score and three subscale scores: physical (nine items), cognitive (10 items), and psychosocial (two items). Responses use a 5-point scale from 0 (never) to 4 (almost always), with higher scores reflecting greater impairment attributed to fatigue. Total scale scores range from 0 to 84. Internal consistency reliability for total and subscale scores ranges from .85 to .97 in this sample.
MS Functional Limitation
Kurtzke’s Incapacity Status Scale (MS-ISS) was used to assess functional limitation related to MS in this study (Kurtzke, 1981). The MS-ISS was developed for individuals with MS and validated by the International Federation of Multiple Sclerosis Societies (Wingerchuk, Noseworthy, & Weinshenker, 1997) and subsequently adapted for use as a self-report survey by Stuifbergen and Roberts (1997). The MS-ISS is a 16-item inventory of functional limitations for role performance and activities of daily living that uses a 5-point scale to rate 16 domains of personal functioning (e.g., ambulation, self-care, and sensory function). A response of 0 indicates no impairment in function, and a response of 4 reflects pronounced or total inability to perform the function. Total scale scores range from 0 to 64, with higher scores reflecting greater functional limitation. Cronbach’s alpha reliability for the MS-ISS in this sample was .87.
Depressive Symptoms
The 10-item short-form Center for Epidemiologic Studies Depression Scale (CES-D) was used to measure depressive symptoms (Andresen, Malmgren, Carter, & Patrick, 1994; Radloff, 1977). The CES-D, which was not developed for use as a diagnostic tool, has been used extensively in epidemiologic studies of healthy and diseased populations. Validity and reliability properties of the CES-D have been established among numerous underserved populations including persons with MS (Verdier-Taillefer, Gourlet, Fuhrer, & Alpe´rovitch, 2001). The short 10-item form of the CES-D has been supported for screening purposes in persons with MS (Kim et al., 2012). The short-form CES-D uses a 5-point scale from 1 (never) to 4 (routinely) to assess depressive symptoms. Total scores range from 10 to 40, with higher scores indicating more depressive symptoms. A cut point of 16 is considered indicative of significant depressive symptoms (Graziano, Calandri, Borghi, & Bonino, 2014). Internal consistency reliability for the 10-item CES-D was .86 in this sample.
Barriers
The Barriers to Health Promoting Activities for Disabled Persons Scale was developed by Becker, Stuifbergen, and Sands (1991) to assess how often specific barriers keep respondents from attending to their health. The 18-item scale incorporates interpersonal, intrapersonal, and environmental barriers (e.g., concern about safety, lack of money). Responses are scaled from 1 (never) to 4 (routinely). Higher scores indicate greater perceived barriers. Internal consistency reliability for this sample was .89.
Personal Resources
The Personal Resource Questionnaire 85 (Part 2; Weinert & Brandt, 1987) was used to measure perceived social support among the respondents. The Personal Resource Questionnaire includes 25 items that are rated from 1 (strongly disagree) to 7 (strongly agree). Higher scores indicate greater perceived support. Content, construct, and criterion-related validity have been supported in the literature (Weinert & Brandt, 1987). Internal consistency reliability for this sample was .93.
Health Promoting Behaviors
The Health Promoting Lifestyle Profile II (HPLP II) was used to assess health promoting behaviors among the respondents (Walker, Sechrist, & Pender, 1987). The HPLP II is a 52-item scale that assesses engagement in activities that support health and well-being.
Responses measure frequency of health promoting behaviors on a 4-point scale (never, sometimes, often, or routinely), with higher scores indicating more frequent practice of health behaviors. Reliability and validity of the HPLP II have been supported in psychometric testing in a large sample of adults (Walker & Hill-Polrecky, 1996). Stuifbergen (1995) showed the use of the HPLP II as a valid measure of health behavior in individuals with chronic disabling conditions including MS. Internal consistency reliability for the HPLP II total score in this sample was .95.
Data Analysis
Statistical analyses were performed using Statistical Package for Social Science, Version 20.0.0, after being proofed for accuracy by research staff. Participants with missing data were excluded from the correlation analysis using pairwise deletion, whereas listwise deletion was used for the regression analyses. Participant characteristics were summarized using descriptive statistics including frequencies, means, and standard deviations. Pearson productYmoment correlations were conducted between age, disease duration, fatigue impact (total, physical, cognitive, and psychosocial), MS-related functional limitation, depressive symptoms, barriers, personal resources, and health promoting behaviors. Four separate standard multiple regression analyses were conducted to predict fatigue impact as conceptualized and operationalized by the MFIS (total, physical, cognitive, and psychosocial fatigue impact). In each analysis, the predictor variables significantly correlated to fatigue (MS-related functional limitation, depressive symptoms, barriers, personal resources, and health promoting behaviors) were entered in one step.
Results
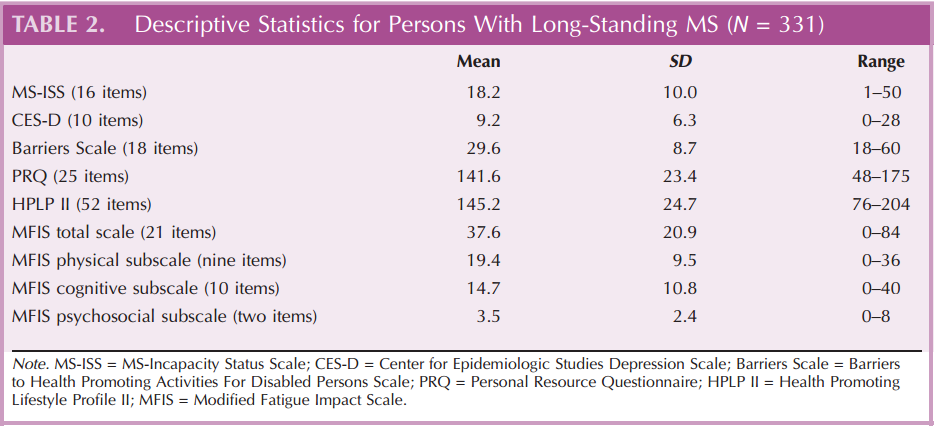
Descriptive statistics (means, standard deviations, and percentiles) were computed for this sample of 331 individuals with long-standing MS (see Table 2). Internal consistency reliability for each of the summated scales exceeded .85.
Pearson’s bivariate correlations denoted that age and MS duration were not significantly (p > .05) related to fatigue impact (see Table 3). Moderate-to-strong positive, linear relationships existed among the fatigueimpact measures and MS functional limitation (range: r = .41–.58, p < .01), depressive symptoms (range: r = .58–.63, p < .01), and barriers (range: r = .55–.63, p < .01). Moderate inverse linear relationships existed among the fatigue impact measures and personal resources (range: r = −.40 to −.50, p < .01) and health promoting behaviors (range: r = −.34 to −.44, p < .01).

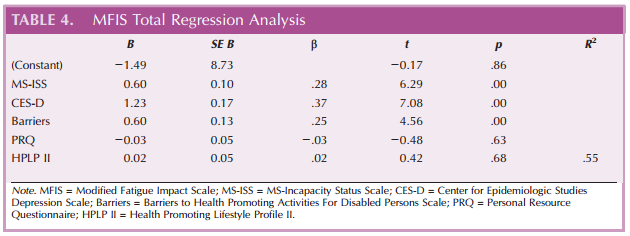
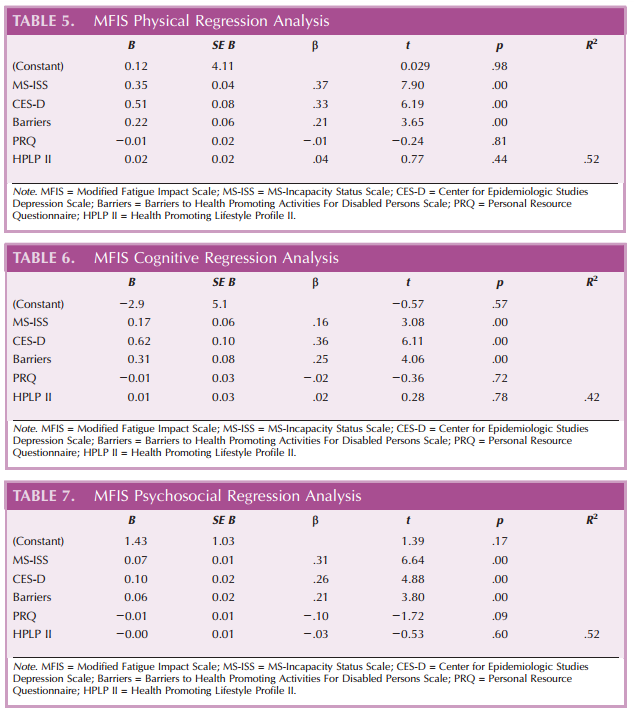
Separate multiple linear regression analyses were performed to predict variation in total, physical, cognitive, and psychosocial fatigue impact. Variables significantly related to fatigue impact were entered simultaneously: MS functional limitation, depressive symptoms, barriers to health promotion, personal resources, and health promoting behaviors. Results showed that all variables in the model, when considered together, explained 54.9% of the variance in total fatigue impact, 52.0% of the variance in physical fatigue impact, 51.7% of the variance in psychosocial fatigue impact, and 41.8% of the variance in cognitive fatigue impact scores (see Tables 4–7). MS functional limitation (MS-ISS) was the strongest predictor in the models for physical fatigue impact (β = .368, p < .01) and psychosocial fatigue impact (β = .308, p < .01), whereas depressive symptoms (CES-D) was the strongest predictor in the models for total fatigue impact (β = .368, p < .01) and cognitive fatigue impact (β = .360, p < .01). Personal resources and health promoting behaviors were not significant predictors of variation for any of the fatigue impact variables.
Discussion
This study was designed to explore relationships among dimensions of fatigue impact in persons with long-standing MS and measures of age, MS duration, MS functional limitation, personal resources, depressive symptoms, barriers to health promoting activities, and health promoting behaviors. Age and MS duration were found to be linearly unrelated to fatigue in this sample. Similarly, Flachenecker et al. (2002) reported that age and disease duration were not related to fatigue measured using either the Modified MS Fatigue Impact Scale (Fisk, Ritvo, et al., 1994) or the Fatigue Severity Scale (Krupp et al., 1989) in a much younger sample of persons with MS (mean age = 39.0 ± 9.3 years) with shorter disease duration (9.9 ± 6.7 years) than the current sample of persons with long-standing MS. This suggests that fatigue may be experienced throughout the disease course irrespective of age or duration of illness.
The models best predicting each of the four dimensions of fatigue impact in the current study included MS-related functional limitation, depressive symptoms, and barriers to health promoting activities. MS functional limitation was found to be the strongest predictor of physical and psychosocial fatigue impact, whereas depressive symptoms was the strongest predictor of total and cognitive fatigue impact in this sample of persons with long-standing MS. Kroencke, Lynch, and Denney (2000) reported that depressed mood and MS disability were significant predictors of fatigue using different instruments (Self-Rating Depression Scale, Expanded Disability Status Scale, and Fatigue Severity Scale) in a younger (mean age = 45.0 + 11.5 years) sample with shorter duration (8.7 ± 8.2 years) of MS (Krupp et al., 1989; Kurtzke, 1983; Zung, 1965). These results uphold the importance of nurses assessing and monitoring depressive symptoms and functional status as well as dimensions of fatigue at each opportunity.
Barriers to health promoting activities was the weakest predictor of total, physical, and psychosocial fatigue impact, whereas functional impairment was the weakest predictor of cognitive fatigue impact. This variation in the relationships between predictors and dimensions of fatigue impact may elucidate modifiable factors for nurses to target in interventions to manage fatigue. Conceptually, it makes sense that MS functional incapacity is predictive of physical and psychosocial fatigue impact. Similarly, it is plausible that depressive symptoms predict cognitive as well as total fatigue impact.
These findings suggest that MS functional impairment, depressive symptoms, and barriers to health promoting activities are associated with fatigue impact in individuals with long-standing MS. Although a positive relationship between functional impairments (e.g., mobility, cognitive) and fatigue in MS has been described in a number of studies including a large (n = 9077) North American registry sample, the mechanisms of the relationship have yet to be explicated (Bol, Duits, Hupperts, Vlaeyen, & Verhey, 2009; Hadjimichael, Vollmer, Oleen-Burkey, & North American Research Committee on Multiple Sclerosis, 2008, Kos, Kerckhofs, Nagels, D’hooghe, & Ilsbroukx, 2008). Significant associations between depression and fatigue in persons with MS have also been reported (Bol et al., 2009; Kroencke et al., 2000). The relationship between fatigue and depression is quite complex and has numerous symptoms in common (e.g., sleep disturbances, low motivation), with causality yet to be explained (Kos et al., 2008). Becker and Stuifbergen (2004) described fatigue and impairment as the most commonly cited barriers to health promoting activities among a sample of persons with MS. Future intervention studies designed to improve management of fatigue should include content related to identification and management of depressive symptoms as well as strategies to address barriers to health promoting activities.
Limitations of this study include a lack of demographic diversity in this predominantly well-educated, White, married, female sample with long-standing MS. The cross-sectional design and correlational analyses prohibit attributing causal means to the findings. In addition, participants are “survivors” in a 17-year longitudinal study and may differ in unknown ways from other groups of persons with long-standing MS. To our knowledge, this is the first study published on fatigue impact where the entire sample is known to have long-standing MS (> 17 years).
MS functional impairment, depressive symptoms, and barriers to health promoting activities may be significant sources of fatigue among persons with long-standing MS. Functional impairment related to pathophysiologic changes in the central nervous system (inflammation and axonal loss) may underlie fatigue in MS, but the exact mechanisms are unclear (Kos et al., 2008; Krupp et al., 2010). Assessment and treatment of depression has the potential to lessen the impact of fatigue in persons with MS (Krupp, 2003). Additional research is needed to explore if interventions to enhance management of depressive symptoms and barriers to health promoting activities would result in lower fatigue impact in persons with long-standing MS.
Acknowledgements
This work is supported in part with grant funding from the National Institutes of Health, National Institute of Nursing Research to the authors (F31NRO14601 and R01NR003195) and by the James R. Dougherty Jr. Centennial Professorship in Nursing at the University of Texas at Austin.
References
Andresen, E. M., Malmgren, J. A., Carter, W. B., & Patrick, D. L. (1994). Screening for depression in well older adults: Evaluation of a short form of the CES-D (Center for Epidemiologic Studies Depression Scale). American Journal of Preventive Medicine, 10(2), 77Y84.
Becker, H., & Stuifbergen, A. (2004). What makes it so hard? Barriers to health promotion experienced by people with multiple sclerosis and polio. Family and Community Health, 27(1), 75Y85.
Becker, H., Stuifbergen, A. K., & Sands, D. (1991). Development of a scale to measure barriers to health promotion activities among persons with disabilities. American Journal of Health Promotion, 5(6), 449Y454.
Bol, Y., Duits, A. A., Hupperts, R. M., Vlaeyen, J. W., & Verhey, F. R. (2009). The psychology of fatigue in patients with multiple sclerosis: A review. Journal of Psychosomatic Research, 66(1), 3Y11. doi:10.1016/j.jpsychores .2008.05.003
Consortium of Multiple Sclerosis Centers Health Services Research Subcommittee. (1997). Multiple sclerosis quality of life inventory: A user’s manual. New York, NY: National Multiple Sclerosis Society. Retrieved from http://www.nationalmssociety. org/ms-clinical-care-network/researchers/clinical-studymeasures/msqli/index.aspx
Fisk, J. D., Pontefract, A., Ritvo, P. G., Archibald, C. J., & Murray, T. J. (1994). The impact of fatigue on patients with multiple sclerosis. Canadian Journal of Neurological Sciences, 21(1), 9Y14.
Fisk, J. D., Ritvo, P. G., Ross, L., Haase, D. A., Marrie, T. J., & Schlech, W. F. (1994). Measuring the functional impact of fatigue: Initial validation of the fatigue impact scale. Clinical Infectious Diseases, 18(Suppl. 1), S79YS83.
Flachenecker, P., K[mpfel, T., Kallmann, B., Gottschalk, M., Grauer, O., Rieckmann, P., I Toyka, K. V. (2002). Fatigue in multiple sclerosis: A comparison of different rating scales and correlation to clinical parameters. Multiple Sclerosis, 8(6), 523Y526.
Graziano, F., Calandri, E., Borghi, M., & Bonino, S. (2014). The effects of a group-based cognitive behavioral therapy on people with multiple sclerosis: A randomized controlled trial. Clinical Rehabilitation, 28(3), 264Y274. doi:10.1177/ 0269215513501525
Hadjimichael, O., Vollmer, T., Oleen-Burkey, M., & North American Research Committee on Multiple Sclerosis. (2008). Fatigue characteristics in multiple sclerosis: The North American Research Committee on Multiple Sclerosis (NARCOMS) survey. Health and Quality of Life Outcomes, 6, 100.
Induruwa, I., Constantinescu, C. S., & Gran, B. (2012). Fatigue in multiple sclerosisVA brief review. Journal of the Neurological Sciences, 323(1Y2), 9Y15. doi:10.1016/j.jns.2012.08.007
Julian, L. J., Vella, L., Vollmer, T., Hadjimichael, O., & Mohr, D. C. (2008). Employment in multiple sclerosis. Exiting and reentering the work force. Journal of Neurology, 255(9), 1354Y1360. doi:10.1007/s00415-008-0910-y
Kim, J., Amtmann, D., Cook, K. F., Johnson, K. L., Bamer, A. M., Chung, H., & Askew, R. L. (2012). Psychometric properties of three depression scales in people with multiple sclerosis. International Journal of MS Care, 14(Suppl. 2), S85YS86.
Kluger, B. M., Krupp, L. B., & Enoka, R. M. (2013). Fatigue and fatigability in neurologic illnesses: Proposal for a unified taxonomy. Neurology, 80(4), 409Y416. doi:10.1212/WNL .0b013e31827f07be
Kos, D., Kerckhofs, E., Nagels, G., D’hooghe, M. B., & Ilsbroukx, S. (2008). Origin of fatigue in multiple sclerosis: Review of the literature. Neurorehabilitation and Neural Repair, 22(1), 91Y100. doi:10.1177/1545968306298934
Kroencke, D. C., Lynch, S. G., & Denney, D. R. (2000). Fatigue in multiple sclerosis: relationship to depression, disability, and disease pattern. Multiple sclerosis, 6, 131Y136.
Krupp, L. B. (2003). Fatigue in multiple sclerosis: Definition, pathophysiology and treatment. CNS Drugs, 17(4), 225Y234.
Krupp, L. B., & Christodoulou, C. (2001). Fatigue in multiple sclerosis. Current Neurology and Neuroscience Reports, 1, 294Y298.
Krupp, L. B., LaRocca, N. G., Muir-Nash, J., & Steinberg, A. D. (1989). The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Archives of Neurology, 46(10), 1121Y1123.
Krupp, L. B., Serafin, D. J., & Christodoulou, C. (2010). Multiple sclerosis-associated fatigue. Expert Review of Neurotherapeutics, 10(9), 1437Y1447. doi:10.1586/ern.10.99
Kurtzke, J. F. (1981). A proposal for a uniform minimal record of disability in multiple sclerosis. Acta Neurologica Scandinavica Supplementum, 64, 110Y129.
Kurtzke, J. F. (1983). Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology, 33(11), 1444Y1452.
Minden, S. L., Frankel, D., Hadden, L., Perloffp, J., Srinath, K. P., & Hoaglin, D. C. (2006). The Sonya Slifka Longitudinal Multiple Sclerosis Study: Methods and sample characteristics. Multiple Sclerosis, 12(1), 24Y38.
Multiple Sclerosis Council for Clinical Practice Guidelines. (1998). Fatigue and multiple sclerosis: Evidence-based management strategies for fatigue in multiple sclerosis. Washington, DC: Paralyzed Veterans of America.
Multiple Sclerosis International Federation. (2013). Atlas of MS 2013. Retrieved from http://www.atlasofms.org
Radloff, L. S. (1977). The CES-D scale: A self-report depression scale for research in the general population. Applied Psychological Measurement, 1, 385Y401. doi:10.1177/ 014662167700100306
Smith, M. M., & Arnett, P. A. (2005). Factors related to employment status changes in individuals with multiple sclerosis. Multiple Sclerosis, 11(5), 602Y609.
Stuifbergen, A. K. (1995). Health-promoting behaviors and quality of life among individuals with multiple sclerosis. Scholarly Inquiry for Nursing Practice, 9(1), 31Y50.
Stuifbergen, A. K., & Roberts, G. J. (1997). Health promotion practices of women with multiple sclerosis. Archives of Physical Medicine and Rehabilitation, 78(12, Suppl. 5), S3YS9.
Verdier-Taillefer, M. H., Gourlet, V., Fuhrer, R., & Alpe´rovitch, A. (2001). Psychometric properties of the Center for Epidemiologic Studies-Depression scale in multiple sclerosis. Neuroepidemiology, 20(4), 262Y267. doi:10.1159/ 000054800
Walker, S. N., & Hill-Polrecky, D. (1996). Psychometric evaluation of the Health-Promoting Lifestyle Profile II. Proceedings of the 1996 Scientific Session of the American Nurses Association’s Council of Nurse Researchers, June 13-14, Washington, DC.
Walker, S. N., Sechrist, K. R., & Pender, N. J. (1987). The Health-Promoting Lifestyle Profile: Development and psychometric characteristics. Nursing Research, 36(2), 76Y81. Weinert, C., & Brandt, P. A. (1987). Measuring social support with the Personal Resource Questionnaire. Western Journal of Nursing Research, 9(4), 589Y602.
Wingerchuk, D. M., Noseworthy, J. H., & Weinshenker, B. G. (1997). Clinical outcome measures and rating scales in multiple sclerosis trials. Mayo Clinic Proceedings, 72(11), 1070Y1079. doi:10.1016/S0025-6196(11)63550-7
Zung, W. W. (1965). A self-rating depression scale. Archives of General Psychiatry, 12, 63Y70.


