Symptom Management and Lifestyle Modifications in Multiple Sclerosis
Why is this important to me?
MS is a chronic disease that is typically diagnosed in early adulthood and lasts for decades. Symptoms are different in each person and change over time. Symptom management and lifestyle modification can improve symptoms and lead to a healthier and longer life.
Who will benefit from reading this study/article?
This article addresses a variety of symptoms, and thus, all people with MS will benefit from reading this article.
What is the objective of this study?
Symptoms of MS may produce a variety of levels of discomfort, pain, and disability and may decrease quality of life and interfere with activities of daily living. Managing your symptoms will improve your overall health and well-being. Managing your symptoms accompanies the use of disease-modifying therapies and is just as important. Because you may experience more than one symptom and becaused symptoms change over time, you should periodically review your symptoms with your healthcare provider to determine which symptoms concern you most. It is also important to be aware that certain symptoms may simultaneously present, and therefore become more intensified. For example, pain, spasticity, and gait problems may occur together. The following symptoms are common in MS and can be effectively managed:
- Fatigue: Fatigue can affect employment, quality of life, and cognitive abilities. By addressing those things that tend to cause fatigue, a healthcare provider can often assist at reducing fatigue levels. Fatigue may also be reduced with exercise, strategies to conserve energy, and behavioral and drug therapy.
- Sensory problems: These include numbness and pain. Pain, which may cluster with cognitive problems, can be neuropathic (caused by damage to the nervous system), inflammatory, secondary to other MS problems, or unrelated to MS. Treatment is guided by the cause of pain.
- Cognitive problems: These problems typically occur later in MS and include difficulties with information-processing speed, memory, and executive function (planning, attention, managing multiple tasks, etc.). Cognitive problems can affect employment, activities of daily living, and adherence to prescribed medications and rehabilitation activities. Treatment involves ruling out other factors that affect cognition and then creating a coping plan that may involve behavioral therapy, physical and mental exercise, and drug therapy.
- Mood disorders: Major depressive disorder and anxiety are common and occur early in the disease course. Therapy may include drug treatment, psychotherapy, and exercise.
- Bladder problems: These are common in later stages of MS and can lead to social isolation. Treatments include proper hydration, antibiotics, bladder training, medication, nerve stimulation, and sometimes catheterization, which are all aimed at achieving continence, improving quality of life, preventing infection, and protecting the kidneys.
- Spasticity: Spasticity can cause pain, posture problems, limited mobility, and other problems. Management of spasticity includes physical therapy and certain medications.
Modifying your lifestyle choices to achieve wellness and reduce vascular risk factors is important for everyone, especially those with a neurological disease such as MS, because such modifications can improve brain health. Brain weight and volume change over time in everyone, however, the loss of brain volume occurs faster in people with MS. Proper diet, exercise, and avoiding smoking contribute to better brain health and are particularly important if you have MS. The following lifestyle considerations are important:
- Weight: Obesity creates chronic, low-grade inflammation and can cause some degree of damage to the brain. Thus, maintaining optimal body weight if you have MS is important.
- Smoking and alcohol use: Tobacco use can decrease the effectiveness of some MS drugs. More studies are needed to understand the interaction between alcohol use and MS, but if you have MS, you are urged to not smoke or consume excessive amounts of alcohol.
- Vitamin D: Low vitamin D is a risk factor for developing MS. Vitamin D supplements can be useful.
- Diet: The digestive system contains many types of helpful bacteria. The bacterial make-up of your digestive system is influenced by what you eat, vitamin D, smoking, and alcohol consumption. A healthy diet that includes vegetables, fish, vitamins A and D, and certain elements is anti-inflammatory and leads to the presence of healthy bacteria in your digestive system. In contrast, unhealthy fats, red meat, high salt, and high sugar can increase inflammation and have a harmful effect on gut bacteria. Thus, consuming a healthy diet creates a state of healthy bacteria and low inflammation that will benefit brain health. A healthy diet is especially important if you have MS.
- Exercise: Exercise improves many MS symptoms, and people with MS should exercise a few times a week if possible.
- Sleep: Sleep disorders are common in MS and can cause fatigue. Good quality sleep is important for brain health, especially if you have MS.
- Temperature control: MS symptoms can worsen in hot or humid weather, and thus, temperature control can improve MS symptoms.
- Stress: People with MS appear to respond abnormally to stress. Although stress cannot be completely eliminated, steps to reduce stress will benefit you if you have MS.
- Medical marijuana: Medical marijuana may improve some MS symptoms. This topic is controversial, and more studies are needed to assess the risks and benefits.
- Mental and social engagement: If you have MS, you should try to be mentally and socially active, which may improve brain health.
- Vascular risk factors: Heart disease, high blood pressure, high cholesterol, and diabetes negatively impact brain health and should be identified and treated if you have MS.
How did the author study this issue?
The author reviewed studies addressing symptom management and lifestyle choices in people with MS.
| SHARE: | |||||
Original Article
Symptom Management and Lifestyle Modifications in Multiple Sclerosis
Continuum
Patricia K. Coyle, MD, FAAN
ABSTRACT
Purpose of Review: This article reviews the rationale and approach to symptom management and lifestyle modifications in multiple sclerosis (MS).
Recent Findings: MS symptoms are important to treat because they affect quality of life and daily activity. Appreciation of cluster symptoms (where one symptom con-
tributes to another), changes over time, and multimodality therapeutic approaches are guiding optimized symptom management. Equally important are lifestyle modifica-
tions that enhance central nervous system reserve and function. These modifications are the foundation for a health maintenance, wellness, and vascular risk factor control program.
Summary: Symptom management and lifestyle modifications are important therapeutic targets to improve the lives of patients with MS.
Continuum (Minneap Minn) 2016;22(3):815–836.
INTRODUCTION
Multiple sclerosis (MS) is a major acquired central nervous system (CNS) disease of young adults and is on the rise. Every patient is affected differently, but each faces the challenge of living with the disease for decades. This article focuses on two topics. The first is an optimized approach to symptom management. Selected major symptoms will be discussed in depth. The second addresses lifestyle modifications to improve general health and CNS reserve. The goal is to have patients with MS lead better, healthier, and longer lives.
APPROACH TO SYMPTOMS
MS is associated with specific symptoms that affect all disease subtypes (Table 7-1). These symptoms produce variable degrees of discomfort, pain, and suffering. They decrease quality of life and interfere with activities of daily living. Optimal symptom management to enhance the well-being of patients with MS is an important complement to optimal disease-modifying therapy. 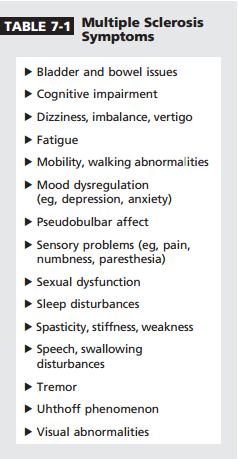
Symptoms strike early. In an online survey of 200 patients with MS aged 35 years or younger, over half reported fatigue (61%), vision disturbances (57%), impaired mobility (55%), dizziness (54%), spasticity (51%), or sleep disturbances (51%).1 MS symptoms evolve over time. In the North American Research Committee on Multiple Sclerosis Global Registry, 11 symptom domains were tracked based on disease duration (from years 0 to 30 after MS onset).2 Each symptom domain was reported by a subset of patients in the first year. All symptoms worsened in some patients over the first decade.
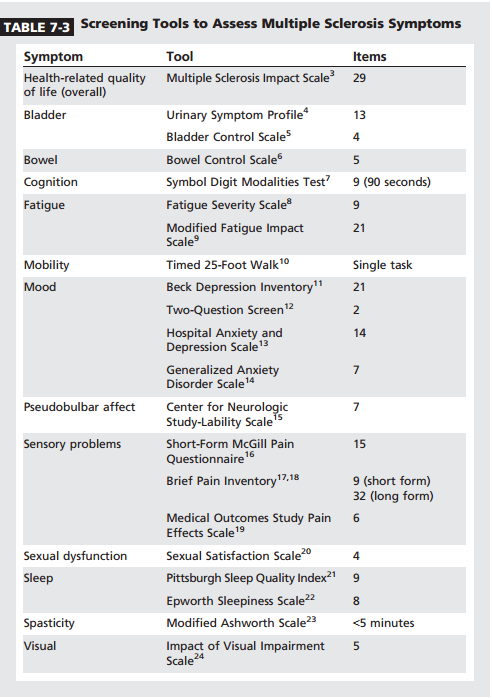
After 15 years, vision, cognitive, sensory, pain, and depression symptoms showed little change. In contrast, mobility, hand function, bladder and bowel, and spasticity symptoms continued to worsen well into the third decade of disease. A basic approach to MS symptom management is outlined in Table 7-2. It emphasizes periodic reassessment of symptoms, since they are moving targets. Ideally this is done at each visit. Patients with MS may have more than one symptom, so it is helpful to have the patient prioritize and list symptoms in rank order of significance. Brief self-report screening forms can be helpful to detect symptoms (Table 7-33Y24). Symptoms often cluster to reinforce one another. One example is pain, spasticity, and gait problems; another is depression, fatigue, and sleep disturbances. Optimal management of one symptom is not likely until its cluster symptoms are effectively treated. With regard to therapy, symptom management is not simply writing a drug prescription; multiple modalities should be considered (Table 7-4).
Fatigue
Fatigue is defined as an overwhelming sense of tiredness. This symptom is subjective and can only be quantified by use of patient-reported outcome scales. Fatigue is often considered the leading MS symptom: 40% of patients say it is their most disabling symptom, and 38% to 83% experience it at some time. Fatigue may occur in the absence of motor dysfunction, is more likely with a mood disorder and a sedentary lifestyle, is recognized to have a major impact on employment (along with cognitive and mobility abnormalities), and impacts quality of life and cognitive functioning (cognitive fatigue becomes apparent with continued efforts). Significant fatigue in isolation may precede a diagnosis of MS by several years.25 Fatigue at the time of a first attack (clinically isolated syndrome) independently predicts a diagnosis of MS.26
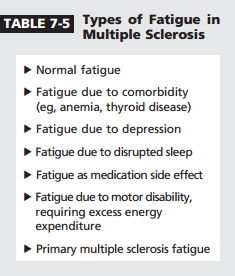
As illustrated in Case 7-1, patients with MS experience different types of fatigue (Table 7-5). The first step in managing fatigue is determining whether it is primary or secondary by reviewing comorbid conditions and medications; checking for depression and sleep issues; and reviewing activity levels, exercise, and use of assistive devices. Primary fatigue (peculiar to MS) involves no precipitating activity, is worsened by heat or humidity (which is not universal, but helpful diagnostically when it occurs), and worsens in the early afternoon. In 30% of patients fatigue may be constant.
The cause of primary MS fatigue is not known. Suggested etiologies include proinflammatory cytokine release (involving interferon gamma; tumor necrosis factor-"; and interleukins 1, 6, and 10); hypothalamicpituitary-adrenal axis dysfunction; axonal injury; cortical atrophy; cerebral lesions involving gray matter; and increased activation of neural circuits, with increased neural excitability.
Proposed therapies for MS fatigue have involved treating comorbid conditions, environmental manipulation (such as use of cooling techniques), regular aerobic and strength training exercises, energy conservation strategies (such as appropriate use of assistive devices), cognitive-behavioral therapy (such as mindfulness), relaxation therapy, magnetic therapy, transcranial stimulation, and drug therapy. Drugs that have been used to treat fatigue include amantadine (100 mg 2 times a day), modafinil (200 mg/d to 400 mg/d), armodafinil (150 mg/d to 250 mg/d), gingko biloba (240 mg/d), and CNS stimulants (both amphetamines and methylphenidate).
 Sensory Problems
Sensory Problems
Sensory problems affect 21% to 55% of patients with early MS and ultimately occur in 52% to 86% of all patients with MS. Sensory issues can be negative (numbness) or positive (paresthesia and pain). Pain (perhaps 63% of sensory problems) is a priority to treat because of associated suffering, but hypesthesia can be of concern if it makes a patient more vulnerable to injury. Pain is typically not prominent early in MS, with the exception of acute neuralgia.
Different types of MS pain exist (Table 7-6). This symptom may cluster with cognitive dysfunction. Acute neuropathic pain involves trigeminal (rarely glossopharyngeal) neuralgia, Lhermitte sign, and dysesthesia. Trigeminal neuralgia affects 1.9% to 6.3% of patients with MS; 90% involve the V2 or V3 distribution.27 No sensory loss occurs, but positive trigger points exist. The American Academy of Neurology has reviewed treatment of trigeminal neuralgia. Objective data exist for carbamazepine (Level A), oxcarbazepine (Level B), and baclofen or lamotrigine (Level C).28 Other medications reported to have benefit include gabapentin, topiramate, and misoprostol. Surgical approaches (eg, percutaneous gasserian ganglion procedures, gamma knife, microvascular decompression) may be considered when medications fail.
 Chronic neuropathic pain is a central syndrome characterized by constant burning pain; it affects 30% to 40% of patients with MS.29 Pain typically involves both lower extremities (worse distally) and occasionally the hands. It is worsened by cold and by deep pressure (cold allodynia, deep hyperesthesia). It is similar to thalamic pain, except that pain is typically unilateral. This syndrome has been associated with lesions in the upper and mid thoracic cord. Treatment involves tricyclic antidepressants, duloxetine (a serotonin norepinephrine reuptake inhibitor), antiepileptic drugs, intrathecal baclofen, cannabinoids (where available), and transcutaneous electrical nerve stimulation. Other MS pain syndromes include acute inflammatory processes (eg, optic neuritis), secondary pain syndromes (eg, due to bladder spasms, musculoskeletal issues, skin breakdown/pressure points, or spasticity), and unrelated pain syndromes (eg, headache; back and joint pain). Treatment is guided by the underlying cause and syndrome. Chronic pain may benefit from the addition of meditation/ mindfulness or biofeedback techniques.
Chronic neuropathic pain is a central syndrome characterized by constant burning pain; it affects 30% to 40% of patients with MS.29 Pain typically involves both lower extremities (worse distally) and occasionally the hands. It is worsened by cold and by deep pressure (cold allodynia, deep hyperesthesia). It is similar to thalamic pain, except that pain is typically unilateral. This syndrome has been associated with lesions in the upper and mid thoracic cord. Treatment involves tricyclic antidepressants, duloxetine (a serotonin norepinephrine reuptake inhibitor), antiepileptic drugs, intrathecal baclofen, cannabinoids (where available), and transcutaneous electrical nerve stimulation. Other MS pain syndromes include acute inflammatory processes (eg, optic neuritis), secondary pain syndromes (eg, due to bladder spasms, musculoskeletal issues, skin breakdown/pressure points, or spasticity), and unrelated pain syndromes (eg, headache; back and joint pain). Treatment is guided by the underlying cause and syndrome. Chronic pain may benefit from the addition of meditation/ mindfulness or biofeedback techniques.
 Cognitive Deficits
Cognitive Deficits
Clear cognitive deficits are unusual early in MS but become more common over time, ultimately occurring in 40% to 70% of all patients. The typical domains involved are information processing speed, memory (working, verbal, and visual), and executive functions. Up to 20% of those affected have sufficient impairment to qualify as having mild dementia. Cognitive deficits affect all MS phenotypes but are typically most notable in secondary progressive MS. However, subtle cognitive disturbances may be identified as early as the radiologically isolated syndrome presymptomatic stage. Cognitive loss has pervasive consequences. It will have a negative impact on employment and activities of daily living, will interfere with therapeutic adherence/compliance and rehabilitation regimens, and may cause marked disability despite preserved motor function. A proportion of patients with so-called benign relapsing MS turn out to have cognitive disability.
The concept of cognitive reserve in MS has been reviewed.30 Cognitive reserve may be defined as the ability to optimize or maximize performance through differential recruitment of brain networks. Baseline cognitive reserve is unique for each individual and contributes to variability. Premorbid cognitive function, tissue destruction and repair, and adaptive functional brain reorganization all play a role. Both genetic (inherited) and environmental (enrichment) factors impact cognitive status and cognitive reserve. In MS, having a larger brain correlates with better cognitive efficiency and less impact of disease burden on cognition.31
MRI findings are associated with cogn itive impairment. The neuropathology of cognitive deficits involves both microscopic and macroscopic injury, cortical plaques, and gray matter volume loss. White matter plaques do not correlate well.32 Functional MRI (fMRI) has detected early disruption in patients with MS, with increased brain circuits required to perform a cognitive task compared to controls.33
Cognitive concerns are subjective about 50% of the time, caused by issues such as depression, anxiety, or disrupted sleep. The gold standard to document objective deficits is full cognitive function testing by a neuropsychologist (which takes several hours), but more limited test batteries can be given over 30 to 90 minutes; some very brief screening tests, such as the Symbol Digit Modalities Test7 and Paced Auditory Serial Addition Test,34 take minutes.
In the patient with MS with cognitive symptoms, it is important to rule out other factors; treat accompanying symptoms that can impact cognition, such as depression, anxiety, pain, fatigue, and sleep disturbance; remove any sedating or anticholinergic drugs; confirm objective deficits with formal testing; and create a coping/rehabilitation plan. Family and significant others need to understand deficits that impact their interactions with the patient.
Treatment of cognitive loss involves cognitive rehabilitation/behavioral therapy (this may be computer assisted training, which may be targeted or nonspecific),35 yoga and exercise, an intellectually enriching lifestyle, and drug therapy. Drug therapy includes stimulants (eg, methylphenidate, amphetamine, modafinil), cholinesterase inhibitors (eg, donepezil, galantamine, rivastigmine), and the N-methyl-D-aspartate (NMDA) receptor antagonist memantine.36 However, a recent Cochrane Review found no convincing data to support any pharmacotherapy.37 Therapy for cognitive deficits is also preventive. It involves effective disease-modifying therapy along with following a health maintenance, wellness, and vascular risk factor control program.
Psychiatric Symptoms/Mood Dysregulation 
Major depressive disorders occur in 36% to 54% of patients with MS, compared to 16.2% of control populations.38 Suicide is at least twice as common in patients with MS. The second most common issue is anxiety disorder, which affects 35.7% of patients with MS, compared to 28.8% of controls.38 Mood disturbance may be seen early. In a 2015 report, depression (particularly anxious depression) was found in 50% of a radiologically isolated syndrome population.39
Depression in MS is multifactorial. It may be reactive, due to structural injury occurring in specific parts of the brain, due to MS-related immune and endocrine changes with hypothalamicpituitary-adrenal axis disruption, or a side effect of certain drug therapies. Alcohol use may be a comorbid factor, and pain is a symptom that may cluster with depression.
Depression features can be divided into those involving mood and those that seem more physical. Depressed mood, loss of interest and pleasure in activities, feelings of guilt and worthlessness, and thoughts of death and suicide are fairly obvious. But increases or decreases in appetite and weight, fatigue and loss of energy, sleep changes (either insomnia or hypersomnia), decreased ability to think and concentrate with indecisiveness, and marked changes in activity level (either psychomotor agitation or retardation) are also signs of depression (Case 7-2). Therapy for depression and anxiety may involve drug treatment, psychotherapy, web-based initiatives, exercise, or other modalities (eg, electroconvulsive therapy, deep brain stimulation, transcranial magnetic stimulation).
 Bladder Dysfunction Lower urinary tract symptoms are unusual in early MS but ultimately occur in 75% of patients. They are associated with pyramidal and spinal cord disease features and an Expanded Disability Status Scale (EDSS) score of 2.0 or higher.40,41 Bladder dysfunction can have a very negative impact on quality of life, including causing social isolation. Patients with MS show three distinct types of neurogenic bladder. The most common form (found in 60% to 80% of patients) is detrusor muscle overactivity (so-called spastic, or overactive, bladder). Small urine volumes trigger inappropriate bladder contraction. Patients report urgency, frequency, urge incontinence, and nocturia. This typically represents a
Bladder Dysfunction Lower urinary tract symptoms are unusual in early MS but ultimately occur in 75% of patients. They are associated with pyramidal and spinal cord disease features and an Expanded Disability Status Scale (EDSS) score of 2.0 or higher.40,41 Bladder dysfunction can have a very negative impact on quality of life, including causing social isolation. Patients with MS show three distinct types of neurogenic bladder. The most common form (found in 60% to 80% of patients) is detrusor muscle overactivity (so-called spastic, or overactive, bladder). Small urine volumes trigger inappropriate bladder contraction. Patients report urgency, frequency, urge incontinence, and nocturia. This typically represents a 
suprapontine or spinal cord lesion.42 The second mo st common neurogenic bladder is detrusor sphincter dyssynergia (in 25%), in which the external sphincter and detrusor are disconnected and simultaneously contract with attempted urination. This represents a suprasacral cord lesion. Patients experience difficulty with initiating urination, double voiding, and incomplete emptying symptoms. The most unusual neurogenic bladder (seen in only 20%) is detrusor hypoactivity (flaccid bladder). This reflects impaired parasympathetic drive from disrupted descending bulbospinal pathways or a sacral cord/conus lesion. The detrusor does not contract at all but fills with urine. Symptoms are decreased or interrupted stream, hesitancy, sensation of incomplete emptying, and dribbling overflow incontinence.
The most feared consequence of neurogenic bladder is upper urinary tract (kidney) involvement. When patients with MS present with bladder issues, recent medications taken should be reviewed; in particular, opiates, antipsychotics, and antidepressants may cause issues. A urinary tract infection must be ruled out. Patients do not always have burning, discoloration, or odor changes with a urinary tract infection. Remedies to combat frequent bladder infections include hydration, vitamin C (to acidify the urine), cranberry juice or cranberry extract (although a recent study of this modality was negative43), using appropriate antibiotics, or even prophylactic antibiotics. The next step is to measure postvoid residual to make sure it is less than 200 mL. Urine volume can be measured by bladder ultrasound. The specific type of neurogenic bladder should be determined, based on symptoms and testing. Occasionally, invasive urodynamics (cystometry and pressure-flow study) are needed for prognostic and therapeutic guidance.
Broad treatment goals involve achieving continence, better quality of life, elimination of bladder infections, and kidney protection,42 starting with diet and lifestyle modifications.44 Patients should be counseled to avoid bladder irritants (eg, alcohol, aspartame, caffeine). Smoking, weight, constipation, and fluid intake issues should be addressed. Bladder training includes scheduled voids, using a Crede´ maneuver or abdominal vibration to try to completely empty the bladder, and pelvic floor or Kegel exercises. Detrusor hyperreflexia generally responds to oral (or patch) medication, including antimuscarinics (anticholinergics), intranasal desmopressin acetate for nocturia (although sodium needs to be monitored), a $3-adrenergic receptor agonist (mirabegron), and cannabinoids. For overactive bladder that fails to respond to oral therapy, intradetrusor injections of onabotulinumtoxinA will last for 10 months. However, bladder infection and temporary urinary retention are recognized complications. Neuromodulation techniques for difficult-to-treat overactive bladder include percutaneous posterior tibial nerve stimulation and sacral neuromodulation (which involves an implantable device that will limit ability to do MRI scans). Bladder instillation of vanilloid receptor agonists, or even oxybutynin, has been proposed but is not in common use. "1-Adrenergic receptor antagonists and antispasticity agents have been used for dyssynergic issues. When a patient has significant retention, catheterization (either intermittent or indwelling) will likely be needed. It is important to note that something can always be done to improve bladder symptoms. To avoid ongoing damage, the sooner neurogenic bladder is addressed, the better.

Spasticity
Spasticity is a velocity-dependent increase in tone that may be generalized or localized. Spasticity may be fixed, leading to contractures, or intermittent, causing spasms. It reflects disrupted supranuclear inhibitory control of spinal interneurons. Ultimately, spasticity affects 60% to 90% of patients with MS.45 When spasticity is mild, it may not require treatment and can even be helpful. In the setting of weakness, spasticity can provide antigravity muscle support to improve ambulation. However, common negative associations include pain, abnormal postures, mobility limitations, disruption in bladder/bowel function, disturbed sleep, pressure sores, and impaired hygiene.
The treatment of spasticity is multifactorial. First, triggers should be corrected or eliminated. Triggers may include a specific joint or extremity positioning, bladder distension, constipation, infection or leg wound, fracture, ingrown toenail, decubitus ulcer, pain, temperature extremes including fever, or tight clothing. Range-ofmotion stretching or physical therapy with structured exercise can improve spasticity for several hours. Other nondrug therapies that have been tried include cryotherapy, transcutaneous electrical stimulation, relaxation, vibration, hydrotherapy, bracing, use of orthotic devices, selective splinting or casting, and noninvasive brain stimulation (repetitive or intermittent theta burst transcranial magnetic stimulation).
Drug therapy for spasticity primarily involves two centrally acting drugs. Baclofen is a ,-aminobutyric acid (GABA)-B receptor agonist that acts at spinal and supraspinal sites. It can be given orally or intrathecally via a surgically implanted programmable pump. Oral baclofen is typically dosed 3 to 4 times a day. Sedation and weakness may occur with higher doses. It should never be stopped abruptly, because a withdrawal syndrome (including seizures, increased spasticity, and hallucinations) may occur. Tizanidine is a central "2-adrenergic agonist that reduces release of excitatory neurotransmitters. It is also typically dosed 3 times a day. Adverse effects include sedation, dizziness, and hypotension. These medications should be started at a low dose, then titrated up. Sometimes combinations of baclofen and tizanidine work well. Other drug therapies include the benzodiazepines (eg, diazepam, clonazepam), which bind to GABA-A receptors; gabapentin; and medical marijuana/cannabinoids (an oral mucosal spray consisting of tetrahydrocannabinol and cannabidiols is approved for spasticity in many countries). Dantrolene is a skeletal muscle relaxant that directly acts on the muscle to block calcium release from the sarcoplasmic reticulum but can be associated with hepatotoxicity. A pilot trial of low-dose naltrexone (4 mg) in primary progressive MS documented significant reduction in spasticity.46 Botulinum toxin injections (which can be guided by ultrasound) are useful for focal limb (such as adductor) spasticity or painful tonic spasms.
As noted earlier, baclofen can be instilled intrathecally with direct access to the lower spinal cord, requiring much lower drug doses. This is an option for severe spasticity when oral regimens are not working or are not tolerated. This is most commonly used in nonambulatory patients but can be helpful in selected ambulatory patients as well. A test bolus of baclofen via lumbar puncture is given first to make sure documented benefit exists before proceeding to surgery. The current battery life of a baclofen pump is about 10 years. It is very rare for other surgical techniques (such as tendon release) to be used to treat MS spasticity in the current era. Spasticity is an important symptom to assess and treat. Patients with MS will need to be reevaluated periodically since it is not a stable symptom.
LIFESTYLE MODIFICATIONS
Following a wellness, health maintenance, and vascular risk factor control program has positive benefits for the CNS. It is important for everyone, but particularly for those with a CNS disease such as MS. Case 7-3 is an example of someone who would benefit from intervention. A wellness program is a universal MS disease modifying therapy.
 Like all organs, the CNS is vulnerable to aging. Brain weight and volume change over time.47 Brain weight increases most markedly up until age 3, then to a lesser extent up until age 18. A decline in brain weight starts in the perimenopause/andropause period, around age 45 to 50 years. Brain volume increases from ages 9 to 13 years, is relatively stable up until age 35, then declines at about 0.2% a year. This annual decline increases to over 0.5% annually after age 60. Brain volume changes reinforce continuous brain plasticity.
Like all organs, the CNS is vulnerable to aging. Brain weight and volume change over time.47 Brain weight increases most markedly up until age 3, then to a lesser extent up until age 18. A decline in brain weight starts in the perimenopause/andropause period, around age 45 to 50 years. Brain volume increases from ages 9 to 13 years, is relatively stable up until age 35, then declines at about 0.2% a year. This annual decline increases to over 0.5% annually after age 60. Brain volume changes reinforce continuous brain plasticity.
Different age-related volume changes exist for gray and white matter. Gray matter segmental volume initially increases in childhood and then starts to decline, while white matter segmental volume increases up to age 45 before starting to decline. In MS, brain volume loss is accelerated and can approximate 1% annually. In a study of 36 pediatric patients with MS aged 5 to 17.7 years, compared to 25 matched controls aged 10.9 to 22 years, the expected brain growth was not seen in the MS cohort; deep gray matter (thalamus) was particularly affected.48 Brain volume loss of 0.1% to 0.3% per year is described as the normal range, with 0.4% or greater annually considered abnormal.
The concept of reserve can be applied broadly to the brain and CNS, in addition to cognition.49 Individual variability encompasses baseline numbers of neurons and synapses, intrinsic abilities, and vulnerability to age- and disease-related changes as well as compensatory mechanisms and plasticity. CNS reserve is amenable to lifestyle changes. Exercise decreases vascular disease, and cognitively stimulating activities decrease hippocampal atrophy, while hypertension, diabetes mellitus, smoking, and obesity are associated with increased brain volume loss. Environmental and life factors can have potent influences on CNS structure and function. This supports the role of lifestyle modifications as an early treatment focus in MS (Table 7-7).
 CNS reserve likely plays a role in the clinical expression of progressive MS. Gradual worsening in MS reflects neurodegeneration (injury to axons and neurons). Onset of progressive MS is age related. In a study of 322 patients with primary progressive MS, 112 patients with single-attack progressive MS, and 421 patients with typical secondary progressive MS, the mean age at progression onset was 45.7, 45.5, and 44.9 years, respectively.50 Neurodegeneration is present from the onset of MS, but clinical expression does not appear until midlife, presumably when sufficient CNS reserve is lost. Core age-related CNS injury factors include a decrease in neuroprotective and repair mechanisms, iron accumulation due to release by oligodendrocytes (with amplification of oxidative injury), and the completion of compact white matter myelination by the fourth decade with subsequent slow white matter tract degeneration
CNS reserve likely plays a role in the clinical expression of progressive MS. Gradual worsening in MS reflects neurodegeneration (injury to axons and neurons). Onset of progressive MS is age related. In a study of 322 patients with primary progressive MS, 112 patients with single-attack progressive MS, and 421 patients with typical secondary progressive MS, the mean age at progression onset was 45.7, 45.5, and 44.9 years, respectively.50 Neurodegeneration is present from the onset of MS, but clinical expression does not appear until midlife, presumably when sufficient CNS reserve is lost. Core age-related CNS injury factors include a decrease in neuroprotective and repair mechanisms, iron accumulation due to release by oligodendrocytes (with amplification of oxidative injury), and the completion of compact white matter myelination by the fourth decade with subsequent slow white matter tract degeneration
Weight and Body Mass Index
Obesity is defined as abnormal or excessive fat accumulation that may impair health.51 The World Health Organization reports worldwide obesity has doubled since 1980. Body mass index (BMI) divides weight by the square of height. BMI over 30 is considered obese and over 25 is considered overweight. This is an epidemic. In 2010, 46% of the American population over age 15 met overweight or obesity criteria, compared to 17% globally. Metabolic syndrome is a disorder of energy utilization and storage, defined by having at least three of five abnormalities: abdominal (central) obesity, high blood pressure, elevated fasting glucose, high triglycerides, and low high-density lipoprotein. Metabolic syndrome increases risk for cardiovascular disease, diabetes mellitus, and stroke.
Obesity is associated with chronic low-grade inflammation, greater brain atrophy and white matter changes, disturbances in the blood-brain barrier, and increased risk for Alzheimer dementia.52 Peripheral white adipose tissue is actually endocrine tissue. It secretes hundreds of adipokines that include cytokines, acute phase reactants, growth factors, and hormones such as leptin, as well as other inflammatory mediators and chemical messengers. Both obesity and metabolic syndrome predispose individuals to inflammatory immune-mediated diseases.
In a 2014 study, 17 very obese but otherwise healthy women scheduled to undergo bariatric surgery were compared to 16 lean women.53 Both groups had cognitive testing and fluorodeoxyglucose positron emission tomography (FDG-PET) brain scans at baseline; studies were repeated in the obese group 6 months postsurgery. At baseline, the obese women showed increased cerebral metabolism in several areas compared to controls, particularly involving the posterior cingulate gyrus (PG.004). Postsurgery, their PET scans normalized. Coincidentally, cognitive executive function performance improved after weight loss surgery.
Obesity/high BMI in childhood, adolescence, and young adulthood is a risk factor for both pediatric and adult-onset MS.54,55 Either clinical mononucleosis or the HLA-DRB1*15 allele further increases MS risk in obese individuals.56,57 This is true for both sexes, but appears more pronounced for females. Achieving optimal body weight is important for anyone with MS.
Tobacco and Alcohol Use
Tobacco is a major public health hazard. It is linked to cancers, cerebrovascular disease, immune-mediated disorders, Alzheimer disease, and neurodevelopmental injury.58 Both active and passive exposure causes injury. In addition to addictive nicotine, tobacco includes over 4000 other chemical compounds. Free radicals, reactive oxygen species, and reactive nitrogen species injure the blood-brain barrier, increase Fas (CD95) expression on lymphocytes to produce a proinflammatory proapoptotic milieu, and increase matrix metalloproteinase. Tobacco, even nicotine-free and ultra-low products, promotes oxidative stress and inflammatory injury.
Tobacco use is a recognized environmental risk factor for the development of MS. It also increases the risk for transition to progressive disease, is associated with cognitive impairment, and is responsible for some of the excess mortality associated with MS.59Y62 Both the duration and intensity of tobacco use are factors, and age does not appear to be a factor. A 2014 study suggested modifications in the NAT1 gene, which is involved in tobacco metabolism, may be linked to the adverse effects of smoking on MS.63 Tobacco use also increases the risk to develop neutralizing antibodies to natalizumab.64 Persistent neutralizing antibodies block efficacy of natalizumab and increase risk for infusion reactions; they are an indication to discontinue natalizumab. Because of the many negative effects of tobacco, all patients with MS should be counseled not to smoke.
Alcohol is a powerful drug. When used in excess, it has clear neurotoxicity. Specific neurologic syndromes are associated with high levels of alcohol as well as with withdrawal after chronic use. Alcohol depresses CNS function and may have additive negative effects when combined with some of the symptomatic pharmacologic treatments for MS. Surprisingly, two Swedish case control studies, with 6619 patients with MS and 7007 controls, found a dosedependent inverse correlation between alcohol use and MS risk.65 This suggests a protective effect of alcohol against developing MS. In contrast, a subsequent record linkage study found a significant positive association between alcohol misuse disorders and MS risk, particularly for men.66 A 2015 review concluded more information was needed to understand whether alcohol use affected risk for MS, risk for disability, and cognitive functioning.67 Patients with MS should avoid excessive alcohol use.
Vitamin D
Vitamin D is a fat-soluble vitamin that is naturally present in or added to certain foods, can be taken as a dietary supplement, and is also produced endogenously by the skin when it is exposed to ultraviolet sun rays. Vitamin D2 is ergocalciferol, and vitamin D3 is cholecalciferol. Vitamin D is inert and must undergo two hydroxylations (by the liver and kidney) to its active form (calcitriol). The best indicator of vitamin D status is serum 25-hydroxy vitamin D (calcidiol) level. Vitamin D promotes intestinal absorption of calcium, iron, magnesium, phosphate, and zinc. It clearly has health benefits beyond just promoting bone growth and remodeling. The cytoplasmic vitamin D receptor affects over 200 genes, many of which are involved in immunity.68 Vitamin D has been implicated in the prevention of cancer, cardiovascular disease, diabetes mellitus, cognitive loss, and immune-mediated disorders.
Low vitamin D is a risk factor for the development of MS.69Y71 Adolescence may be a key time to try to supplement vitamin D.72 A 2014 study suggested that vitamin D levels in early MS predicted clinical and MRI disease activity.73 High levels were associated with less activity than low levels. Low levels have also been associated with greater disability.68 Vitamin D 25-hydroxy levels can be checked annually in patients with MS and replaced to levels of about 60 ng/mL with supplementation of up to 10,000 units daily, barring no contraindication (eg, hypercalcemia or calcium kidney stones).
Diet
Diet influences mucosal immunity. The gut microbiome consists of up to 1014 total bacteria. Every person has at least 100 to 150 unique species. The human gut microbiome has a profound effect on the systemic and mucosal immune systems and is implicated in a variety of disorders, including obesity, cardiovascular disease, inflammatory bowel disease, and immune-mediated disorders such as MS. The gut flora are influenced by diet, vitamin D deficiency, smoking, and alcohol use.74 Gut flora changes may actually lead to intestinal inflammation, altered immunity, decrease in T-regulatory cells, and lipopolysaccharide production with damage to the intestinal barrier and endotoxemia.
Food interacts with cell enzymes, transcription factors, and nuclear receptors to influence inflammation.75 For example, saturated fatty acids activate key inflammatory transcription factors. A diet can be proinflammatory or anti-inflammatory (Table 7-8). High caloric intake, particularly involving refined carbohydrates and sugars, increases insulin levels and increases proinflammatory molecules. In contrast, caloric restriction decreases oxidative injury and lymphocyte activation. Better diets are associated with less cognitive decline.76
There has been recent interest in the effects of dietary salt. Increased salt intake is associated with immune disturbances, including promotion of pathogenic T-helper 17 cells. Higher salt intake in MS was associated with higher relapse rate and MRI lesion activity.77 No proven MS diet exists. Nutrition trials are limited but suggest marginal benefit from linoleic acid and omega 3 and omega 6 polyunsaturated fatty acids. Patients with MS may benefit from a healthy low-cholesterol and low-fat anti-inflammatory diet that minimizes excess proinflammatory factors such as salt.
Exercise
Physical exercise is health promoting for all individuals. It helps to control weight; reduces risk for heart disease, stroke, type 2 diabetes mellitus, metabolic syndrome, and some cancers; improves bone and muscle health; improves mental health, mood, and ability to perform activities of daily living; and increases lifespan. The molecular targets of exercise are identified; they upregulate oxidative metabolism and downregulate biosynthetic pathways and inflammation.75 Exercise increases anti-inflammatory (regulatory) cytokines, decreases plasma leptin and gene expression of its receptors, and increases adiponectin and its receptor. When physical exercise is combined with calorie restriction, marked reduction in inflammatory biomarkers occurs. In animal models, exercise has a positive benefit on brain mitochondrial activity, neuroplasticity, and mood.
Sometimes concerns are raised about exercise worsening MS with prior deficits, similar to postpolio syndrome. A review of 26 randomized controlled trials found no detrimental effects of exercise on relapse rate and no increase in adverse events.78 Exercise improves certain MS symptoms, prevents complications, and may have a neuroprotective component.79 In animal models, exercise increases brain-derived neurotrophic factor, synaptic density, and hippocampal growth. Exercise in patients with MS has improved muscle strength, mobility, and spasticity; fatigue; depression; cognition; and quality of life.80,81 In a randomized controlled trial involving 42 patients with progressive MS treated with three distinct exercise interventions over 8 to 10 weeks (arm ergometry, rowing, bicycle ergometry) versus waitlist control, exercise was associated with significant improvement not only in aerobic fitness, but also walking ability, depression, fatigue, and cognition.82
Exercise can be divided into aerobic and resistance training; both are beneficial for patients with MS. Various exercise proposals for patients with MS have suggested aerobic exercise 3 times a week for 40 minutes; 10 to 40 minutes of aerobics 2 to 3 times a week in combination with resistance training involving 8 to 15 repetitions per set, three to four sets per exercise, 2 days a week; or 30 minutes of aerobics 2 times a week combined with resistance training of 10 to 15 repetitions per set, two sets per exercise, 2 days a week. Whatever plan is followed, the goal for patients with MS is to participate in regular exercise several times a week.
Sleep Hygiene
Sleep allows memory consolidation and promotes normal brain functioning.83 Disrupted sleep is linked to poor health, poor job performance, and increased stress. It disrupts the immune system, causes neuro inflammation, disrupts circadian rhythm and the hypothalamic-pituitary-adrenal axis, interferes with sympathetic activity, and increases risk for diseases. It also causes greater susceptibility to viral infection. Sleep has a strong impact on cognition. Disturbed sleep decreases synaptic plasticity and negatively affects neurogenesis and neuronal pathways.
Sleep disorders are increased in MS and are a recognized cause of fatigue. In a review of 18 studies, the prevalence of obstructive sleep apnea was 7.14% to 58.1%, restless leg syndrome 14.4% to 57.5%, rapid eye movement (REM) behavior disorder 2.22% to 3.2%, and narcolepsy 0% to 1.6%.84 The prevalence of restless legs, for example, was 4 times that of the general population. Sleep hygiene is important for MS not only to avoid disruptive symptoms, but also to enhance CNS health; it should be optimized in MS.
Temperature Control
Patients with MS often note worsening of symptoms in hot or humid weather. This is the basis for the historical diagnostic “hot tub test” and for Uhthoff phenomenon (visual blurring with exercise). The mechanism is believed to be based on ion channel changes on demyelinated axons that lead to transient conduction deficits due to thermoregulatory derangements.
Patients with MS who are heat sensitive can use specific cooling techniques to manipulate their environment. This may include cold drinks, lightweight clothing, fans or air conditioning, cool suits (shirts that attach to cool systems), ice or gel packs, and cooling devices such as collars or vests.
Stress
Psychological stress activates neuroendocrine pathways and alters immune responses. It disrupts both the CNS and the immune system. Chronic stress actually produces changes in peripheral monocytes to increase proinflammatory gene expression.85 Stress activates fear and threat appraisal brain circuitry that produces neurobiological and behavioral responses and activates microglia.
Patients with MS appear to respond to stress abnormally. In a 2014 study, the hypothalamic-pituitary-adrenal axis response to a standardized psychosocial laboratory stressor was significantly increased in the first year of MS and significantly decreased in those with longer disease duration.86 Multiple studies have shown a consistent relationship between psychological stress and MS activity.87
Stress is impossible to eliminate. However, preliminary evidence indicates interventions to reduce stress may benefit MS. These involve mindfulness therapy and stress management programs (eg, relaxation training, meditation, cognitive-behavioral strategies).
Medical Marijuana
Medical marijuana is a controversial topic. A 2014 systematic review of the literature showed benefits of marijuana in treating MS spasticity, pain (central pain and painful spasms), and overactive bladder (Table 7-9).88 An oral spray and two oral capsule forms of marijuana have been marketed globally. As more state legislatures in the United States approve medical marijuana, oral capsule and spray forms will become available to patients with MS. These forms should be encouraged over smoking forms. More research is needed to define the potential risks (particularly cognitive) and benefits of medical marijuana.89
 Mental and Social Stimulation
Mental and Social Stimulation
Lifestyle interventions are recognized to be able to enhance brain function. In essence, they involve environmental enrichment. Mental and social engagement are associated with longer lifespan, better health, better cognition, and less depression. They keep the brain healthier. Social isolation increases morbidity and mortality risks.90
Social and intellectual stimulation appear to boost neurogenesis and dendritic growth and upregulate factors such as brain-derived neurotrophic factor. Life experiences may prevent or minimize brain pathology and may improve cognitive reserve. Patients with MS who are mentally more active show better cognitive function and less brain atrophy than those who are not. Patients with MS with greater early life cognitive leisure activities showed larger hippocampal volumes.91 These leisure activities involved reading (books, magazines, newspapers), producing art (painting, poetry, sculptures), writing (newsletter, diary, essays), playing a musical instrument, playing structured games (crossword, puzzles, cards, board games), and participating in hobbies (model building, gardening, web design). In MS, intellectual enrichment is associated with functional brain circuit changes, including greater brain default network activation and less need for task-active cortical recruitment.30 This MS cognitive reserve model modulates the effect of brain disease burden on cognitive loss. The data support roles for intrinsic and life experience factors in MS cognitive reserve. Thus, it is important for patients with MS to stay mentally and socially active.
Vascular Risk Factors
Vascular risk factors, including cardiac/ heart disease, hypertension, diabetes mellitus, and dyslipidemia, are associated with structural and functional brain injury. In contrast, greater cardiovascular fitness was associated with less brain volume loss and greater white matter integrity 5 years later.92 In a 2014 review of MS and vascular comorbidities, type 2 diabetes mellitus and hypertension appeared to increase disability in MS.93 Cardiovascular disease may be increased in MS and is associated with greater disability; patients with MS are at increased risk to die of cardiovascular causes.
Lipid profiles have also been associated with MS disease course. More severe disease has been associated with increases in total cholesterol, apolipoprotein B, apolipoprotein B to apolipoprotein A-I ratio, and total cholesterol to high-density lipoprotein ratio.94 Vascular risk factors should be identified and treated in patients with MS.
CONCLUSION
Optimized symptom management and lifestyle modifications are two areas that may improve the lives of patients with MS. These should be major focus areas for all neurologists who care for patients with MS.
REFERENCES
1. Patti F, Vila C. Symptoms, prevalence and impact of multiple sclerosis in younger patients: a multinational survey. Neuroepidemiology 2014;42(4):211Y218. doi:10.1159/000360423.
2. Kister I, Bacon TE, Chamot E, et al. Natural history of multiple sclerosis symptoms. Int J MS Care 2013;15(3):146Y158. doi:10.7224/1537-2073.2012-053.
3. Hobart J, Lamping D, Fitzpatrick R, et al. The Multiple Sclerosis Impact Scale (MSIS-29): A new patient-based outcome measure. Brain 2001;124(pt 5):962Y973.
4. Haab F, Richard F, Amarenco G, et al. Comprehensive evaluation of bladder and urethral dysfunction symptoms: development and psychometric validation of the Urinary Symptom Profile (USP) questionnaire. Urology 2008;71(4):646Y656. doi:10.1016/ j.urology.2007.11.100.
5. Bladder Control Scale (BLCS). National Multiple Sclerosis Society website. www. nationalmssociety.org/For-Professionals/ Researchers/Resources-for-Researchers/ Clinical-Study-Measures/Bladder-ControlScale-(BLCS). Accessed March 30, 2016.
6. Bowel Control Scale (BWCS). National Multiple Sclerosis website. www. nationalmssociety.org/For-Professionals/ Researchers/Resources-for-Researchers/ Clinical-Study-Measures/Bowel-ControlScale-(BWCS). Accessed March 30, 2016.
7. Smith A. Symbol Digit Modalities Test. Los Angeles, CA: Western Psychological Services; 1982.
8. Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD. The fatigue severity scale: application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 1989;46(10):1121Y1123.
9. Modified Fatigue Impact Scale (MFIS). National Multiple Sclerosis Society website. www.nationalmssociety.org/ForProfessionals/Researchers/Resources-forResearchers/Clinical-Study-Measures/ Modified-Fatigue-Impact-Scale-(MFIS). Accessed March 30, 2016.
10. Timed 25-Foot Walk (T25-FW). National Multiple Sclerosis Society website. www. nationalmssociety.org/For-Professionals/ Researchers/Resources-for-Researchers/ClinicalStudy-Measures/Timed-25-Foot-Walk- (T25-FW). Accessed March 30, 2016.
11. Beck AT, Ward CH, Mendelson M, et al. An inventory for measuring depression. Arch Gen Psychiatry 1961;4(6):561Y571.
12. Two question screen. www.uphp.com/wpcontent/uploads/2013/10/TwoQuestion Screen.pdf. Published December 4, 2002. Accessed March 30, 2016.
13. Hospital Anxiety and Depression Scale. www.scalesandmeasures.net/files/files/ HADS.pdf. Accessed April 6, 2016.
14. Generalized Anxiety Disorder 7-item (GAD-7) scale. www.integration.samhsa.gov/ clinical-practice/GAD708.19.08Cartwright.pdf. Accessed March 30, 2016.
15. Moore SR, Gresham LS, Bromberg MB, et al. A self report measure of affective lability. J Neurol Neurosurg Psychiatry 1997;63(1):89Y93.
16. Short Form McGill Pain Questionnaire. www. npcrc.org/files/news/mcgill_pain_inventory. pdf. Accessed March 30, 2016.
17. Brief Pain Inventory (Short Form). www. npcrc.org/files/news/briefpain_short.pdf. Published 1991. Revised July 1, 2005. Accessed March 30, 2016.
18. The Brief Pain Inventory. www.npcrc.org/ files/news/briefpain_long.pdf. Published 1991. Accessed March 30, 2016.
19. MOS Pain Effects Scale (PES). National Multiple Sclerosis Society website. www. nationalmssociety.org/For-Professionals/ Researchers/Resources-for-Researchers/ClinicalStudy-Measures/MOS-Pain-Effects-Scale-(PES). Accessed March 30, 2016.
20. Sexual Satisfaction Scale (SSS). National Multiple Sclerosis website. www. nationalmssociety.org/For-Professionals/ Researchers/Resources-for-Researchers/ Clinical-Study-Measures/Sexual-SatisfactionScale-(SSS). Accessed March 30, 2016.
21. Buysse DJ, Reynolds III CF, Monk TH, et al. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res 1989; 28(2):193Y213.
22. Epworth Sleepiness Scale. epworthsleepinessscale.com/epworthsleepiness-scale.pdf. Accessed March 30, 2016.
23. Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther 1987;67(2): 206Y207.
24. Impact of Visual Impairment Scale (IVIS). National Multiple Sclerosis Society website. www.nationalmssociety.org/ForProfessionals/Researchers/Resources-forResearchers/Clinical-Study-Measures/Impact-ofVisual-Impairment-Scale-(IVIS). Accessed March 30, 2016.
25. Berger JR, Pocoski J, Preblick R, Boklage S. Fatigue heralding multiple sclerosis. Mult Scler 2013;19(11):1526Y1532. doi:10.1177/ 1352458513477924.
26. Runia TF, Jafari N, Siepman DA, Hintzen RQ. Fatigue at a time of CIS is an independent predictor of a subsequent diagnosis of multiple sclerosis. J Neurol Neurosurg Psychiatry 2015;86(5):543Y546. doi:10.1136/ jnnp-2014-308374.
27. Solaro C, Trabucco E, Messmer Uccelli M. Pain and multiple sclerosis: pathophysiology and treatment. Curr Neurol Neurosci Rep 2013;13(1):320. doi:10.1007/s11910-012-0320-5.
28. Gronseth G, Cruccu G, Alksne J, et al. (2008) Practice parameter: the diagnostic evaluation and treatment of trigeminal neuralgia (an evidence-based review): a report of the Quality Standards Subcommittee of the American Academy of Neurology and the European Federation of Neurological Societies. Neurology 2008;71(15):1183Y1190. doi:10.1212/01.wnl.0000326598.83183.04.
29. Okuda DT, Melmed K, Matsuwaki T, et al. Central neuropathic pain in MS is due to distinct thoracic spinal cord lesions. Ann Clin Transl Neurol 2014;1(8):554Y561. doi:10.1002/acn3.85.
30. Sumowski JF, Leavitt VM. Cognitive reserve in multiple sclerosis. Mult Scler 2013;19(9): 1122Y1127. doi:10.1177/1352458513498834.
31. Sumowski JF. Cognitive reserve as a useful concept for early intervention research in multiple sclerosis. Front Neurol 2015;6:176. doi:10.3389/fneur.2015.00176.
32. Rocca MA, Amato MP, De Stefano N, et al. Clinical and imaging assessment of cognitive dysfunction in multiple sclerosis. Lancet Neurol 2015;14(3):302Y317. doi:10.1016/ S1474-4422(14)70250-9.
33. Staffen W, Mair A, Zaune H, et al. Cognitive function and fMRI in patients with multiple sclerosis: evidence for compensatory cortical activation during an attention task. Brain 2002;125(pt 6):1275Y1282.
34. Wiens AN, Fulle KH, Crossen JR. Paced Auditory Serial Addition Test: adult norms and moderator variables. J Clin Exp Neuropsychol 1997;19(4):473Y483.
35. Bonavita S, Sacco R, Della Corte M, et al. Computer-aided cognitive rehabilitation improves cognitive performances and induces brain functional connectivity changes in relapsing remitting multiple sclerosis patients: an exploratory study. J Neurol 2015;262(1):91Y100. doi:10.1007/ s00415-014-7528-z.
36. Dresler M, Sandberg A, Ohla K, et al. Non-pharmacological cognitive enhancement. Neuropharmacology 2013;64:529Y543. doi:10.1016/j.neuropharm.2012.07.002.
37. He D, Zhang Y, Dong S, et al. Pharmacological treatment for memory disorder in multiple sclerosis. Cochrane Database Syst Rev 2013;12:CD008876. doi:10.1002/14651858.CD008876.pub3.
38. Minden SL, Feinstein S, Kalb RC, et al. Evidence-based guideline: assessment and management of psychiatric disorders in individuals with MS: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology 2014;82(2):174Y181. doi:10.1212/ WNL.0000000000000013.
39. Labiano-Fontcuberta A, Aladro Y, Martı´nezGine´s ML, et al. Psychiatric disturbances in radiologically isolated syndrome. J Psychiatr Res 2015;68:309Y315. doi:10.1016/ j.jpsychires.2015.05.008.
40. Ghezzi A, Mutta E, Bianchi F, et al. Diagnostic tools for assessment of urinary dysfunction in MS patients without urinary disturbances (published online ahead of print November 27, 2015). Neurol Sci. doi:10.1007/s10072-015-2415-7.
41. Fox RJ, Bacon TE, Chamot E, et al. Prevalence of multiple sclerosis symptoms across lifespan: data from the NARCOMS Registry. Neurodegener Dis Manag 2015;5(6 suppl): 3Y10. doi:10.2217/nmt.15.55.
42. Panicker JN, Fowler CJ, Kessler TM. Lower urinary tract dysfunction in the neurological patient: clinical assessment and management. Lancet Neurol 2015;14(7): 720Y732. doi:10.1016/S1474-4422(15)00070-8.
43. Gallien P, Amarenco G, Benoit N, et al. Cranberry versus placebo in the prevention of urinary infections in multiple sclerosis: a multicenter, randomized, placebo-controlled, double-blind trial. Mult Scler 2014;20(9): 1252Y1259. doi:10.1177/1352458513517592.
44. Sadiq A, Brucker BM. Management of neurogenic lower urinary tract dysfunction in multiple sclerosis patients. Curr Urol Rep 2015;16(7):44. doi:10.1007/ s11934-015-0519-5.
45. Feinstein A, Freeman J, Lo AC. Treatment of progressive multiple sclerosis: what works, what does not, and what is needed. Lancet Neurol 2015;14(2):194Y207. doi:10.1016/ S1474-4422(14)70231-5.
46. Gironi M, Martinelli-Boneschi F, Sacerdote P, et al. A pilot trial of low-dose naltrexone in primary progressive multiple sclerosis. Mult Scler 2008;14(8):1076Y1083. doi:10.1177/1352458508095828.
47. Hedman AM, van Haren NE, Schnack HG, et al. Human brain changes across the life span: a review of 56 longitudinal magnetic resonance imaging studies. Hum Brain Mapp 2012;33(8):1987Y2002. doi:10.1002/hbm.21334.
48. Aubert-Broche B, Fonov V, Narayanan S, et al. Onset of multiple sclerosis before adulthood leads to failure of age-expected brain growth. Neurology 2014;83(23): 2140Y2146. doi:10.1212/WNL. 0000000000001045.
49. Stern Y. Cognitive reserve in ageing and Alzheimer’s disease. Lancet Neurol 2012;11(11):1006Y1012. doi:10.1016/S1474- 4422(12)70191-6.
50. Tutuncu M, Tang J, Zeid NA, et al. Onset of progressive phase is an age-dependent clinical milestone in multiple sclerosis. Mult Scler 2013;19(2):188Y198. doi:10.1177/ 1352458512451510.
51. Manzel A, Muller DN, Hafler DA, et al. Role of “Western diet” in inflammatory autoimmune diseases. Curr Allergy Asthma Rep 2014;14(1):404. doi:10.1007/ s11882-013-0404-6.
52. Kiliaan AJ, Arnoldussen IA, Gustafson DR. Adipokines: a link between obesity and dementia? Lancet Neurol 2014;13(9): 913Y923. doi:10.1016/S1474-4422(14)70085-7.
53. Marques EL, Halpern A, Correˆa Mancini M, et al. Changes in neuropsychological tests and brain metabolism after bariatric surgery. J Clin Endocrinol Metab 2014;99(11): E2347YE2352. doi:10.1210/jc.2014-2068.
54. Wesnes K, Riise T, Casetta I, et al. Body size and the risk of multiple sclerosis in Norway and Italy: the EnvIMS Study. Mult Scler 2015;21(4):388Y395. doi:10.1177/ 1352458514546785.
55. Kavak KS, Teter BE, Hagemeier J, et al. Higher weight in adolescence and young adulthood is associated with an earlier age at multiple sclerosis onset. Mult Scler 2015;21(7):858Y865. doi:10.1177/ 1352458514555787.
56. Hedstro¨ m AK, Lima Bomfim I, Hillert J, et al. Obesity interacts with infectious mononucleosis in risk of multiple sclerosis. Eur J Neurol 2015;22(3):578Ye38. doi:10.1111/ene.12620.
57. Hedstro¨ m AK, Lima Bomfim I, Barcellos L, et al. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology 2014;82(10):865Y872. doi:10.1212/WNL.0000000000000203.
58. Naik P, Fofaria N, Prasad S, et al. Oxidative and pro-inflammatory impact of regular and denicotinized cigarettes on blood brain barrier endothelial cells: is smoking reduced or nicotine-free products really safe? BMC Neurosci 2014;15:51. doi:10.1186/1471- 2202-15-51.
59. Hedstro¨ m AK, Hillert J, Olsson T, Alfredsson L. Smoking and multiple sclerosis susceptibility. Eur J Epidemiol 2013;28(11):867Y874. doi:10.1007/s10654-013-9853-4.
60. Manouchehrinia A, Tench CR, Maxted J, et al. Tobacco smoking and disability progression in multiple sclerosis: United Kingdom cohort study. Brain 2013;136(pt 7): 2298Y2304. doi:10.1093/brain/awt139.
61. Ozcan ME, Ince B, Bingo¨ l A, et al. Association between smoking and cognitive impairment in multiple sclerosis. Neuropsychiatr Dis Treat 2014;10:1715Y1719. doi:10.2147/NDT.S68389.
62. Manouchehrinia A, Weston M, Tench CR, et al. Tobacco smoking and excess mortality in multiple sclerosis: a cohort study. J Neurol Neurosurg Psychiatry 2014;85(10):1091Y1095. doi:10.1136/jnnp-2013-307187.
63. Briggs FB, Acuna B, Shen L, et al. Smoking and risk of multiple sclerosis: evidence of modification by NAT1 variants. Epidemiology 2014;25(4):605Y614. doi:10.1097/ EDE.0000000000000089.
64. Hedstro¨ m A, Alfredsson L, Lundkvist Ryner M, et al. Smokers run increased risk of developing anti-natalizumab antibodies. Mult Scler 2013;20(8):1081Y1085. doi:10.1177/1352458513515086.
65. Hedstro¨ m AK, Hillert J, Olsson T, Alfredsson L. Alcohol as a modifiable lifestyle factor affecting multiple sclerosis risk. JAMA Neurol 2014;71(3):300Y305. doi:10.1001/ jamaneurol.2013.5858.
66. Pakpoor J, Goldacre R, Disanto G, et al. Alcohol misuse disorders and multiple sclerosis risk. JAMA Neurol 2014;71(9): 1188Y1189. doi:10.1001/jamaneurol. 2014.1795.
67. Wang YJ, Li R, Yan JW, et al. The epidemiology of alcohol consumption and multiple sclerosis: a review. Neurol Sci 2015;36(2):189Y196. doi:10.1007/s10072-014-2007-y.
68. Thouvenot E, Orsini M, Daures JP, Camu W. Vitamin D is associated with degree of disability in patients with fully ambulatory relapsing-remitting multiple sclerosis. Eur J Neurol 2015;22(3):564Y569. doi:10.1111/ ene.12617.
69. Burton JM, Costello FE. Vitamin D in multiple sclerosis and central nervous system demyelinating diseaseVa review. J Neuroophthalmol 2015;35(2):194Y200. doi:10.1097/WNO.0000000000000256.
70. Correale J, Gaita´n MI. Multiple sclerosis and environmental factors: the role of vitamin D, parasites, and Epstein-Barr virus infection. Acta Neurol Scand Suppl 2015;132(199): 46Y55. doi:10.1111/ane.12431.
71. Sundstro¨ m P, Salzer J. Vitamin D and multiple sclerosisVfrom epidemiology to prevention. Acta Neurol Scand Suppl 2015;132(199):56Y61. doi:10.1111/ane.12432.
72. Cortese M, Riise T, Bjørnevik K, et al. Timing of use of cod liver oil, a vitamin D source, and multiple sclerosis risk: the EnvIMS study. Mult Scler 2015. doi:10.1177/ 1352458515578770.
73. Ascherio A, Munger KL, White R, et al. Vitamin D as an early predictor of multiple sclerosis activity and progression. JAMA Neurol 2014;71(3):306Y314. doi:10.1001/ jamaneurol.2013.5993.
74. Mielcarz DW, Kasper LH. The gut microbiome in multiple sclerosis. Curr Treat Options Neurol 2015;17(4):344. doi:10.1007/ s11940-015-0344-7.
75. Riccio P, Rossano R. Nutrition facts in multiple sclerosis. ASN Neuro 2015;7(1):pii: 1759091414568185. doi:10.1177/ 1759091414568185.
76. Smyth A, Dehghan M, O’Donnell M, et al. Healthy eating and reduced risk of cognitive decline: a cohort from 40 countries. Neurology 2015;84(22):2258Y2265. doi:10.1212/WNL.0000000000001638.
77. Farez MF, Fiol MP, Gaita´n MI, et al. Sodium intake is associated with increased disease activity in multiple sclerosis. J Neurol Neurosurg Psychiatry 2015;86(1):26Y31. doi:10.1136/ jnnp-2014-307928.
78. Pilutti LA, Platta ME, Motl RW, LatimerCheung AE. The safety of exercise training in multiple sclerosis: a systematic review. J Neurol Sci 2014;343(1Y2):3Y7. doi:10.1016/ j.jns.2014.05.016.
79. Giesser BS. Exercise in the management of persons with multiple sclerosis. Ther Adv Neurol Disord 2015;8(3):123Y130. doi:10.1177/1756285615576663.
80. Mulero P, Almansa R, Neri MJ, et al. Improvement of fatigue in multiple sclerosis by physical exercise is associated to modulation of systemic interferon response. J Neuroimmunol 2015;280:8Y11. doi:10.1016/j.jneuroim.2015.01.011.
81. Adamson BC, Ensari I, Motl RW. Effect of exercise on depressive symptoms in adults with neurologic disorders: a systematic review and meta-analysis. Arch Phys Med Rehabil 2015;96(7):1329Y1338. doi:10.1016/j.apmr.2015.01.005.
82. Briken S, Gold SM, Patra S, et al. Effects of exercise on fitness and cognition in progressive MS: a randomized, controlled pilot trial. Mult Scler 2014;20(3):382Y390. doi:10.1177/ 1352458513507358.
83. Yaffe K, Falvey CM, Hoang T. Connections between sleep and cognition in older adults. Lancet Neurol 2014; 13(10):1014Y1028. doi:10.1016/ S1474-4422(14)70172-3.
84. Marrie RA, Reider N, Cohen J, et al. A systematic review of the incidence and prevalence of sleep disorders and seizure disorders in multiple sclerosis. Mult Scler 2015;21(3):342Y349. doi:10.1177/ 1352458514564486.
85. Wohleb ES, McKim DB, Sheridan JF, Godbout JP. Monocyte trafficking to the brain with stress and inflammation: a novel axis of immune-to-brain communication that influences mood and behavior. Front Neurosci 2015;8:447. doi:10.3389/ fnins.2014.00447.
86. Kern S, Rohleder N, Eisenhofer G, et al. Time mattersVacute stress response and glucocorticoid sensitivity in early multiple sclerosis. Brain Behav Immun 2014;41:82Y89. doi:10.1016/j.bbi.2014.04.006.
87. Lovera J, Reza T. Stress in multiple sclerosis: review of new developments and future directions. Curr Neurol Neurosci Rep 2013;13(11):398. doi:10.1007/ s11910-013-0398-4.
88. Koppel BS, Brust JC, Fife T, et al. Systematic review: efficacy and safety of medical marijuana in selected neurologic disorders: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology 2014;82(17): 1556Y1563. doi:10.1212/WNL. 0000000000000363.
89. Pavisian B, MacIntosh BJ, Szilagyi G, et al. Effects of cannabis on cognition in patients with MS: a psychometric and MRI study. Neurology 2014;82(21):1879Y1887. doi:10.1212/WNL.0000000000000446.
90. Cho HJ, Seeman TE, Kiefe CI, et al. Sleep disturbance and longitudinal risk of inflammation: moderating influences of social integration and social isolation in the Coronary Artery Risk Development in Young Adults (CARDIA) study. Brain Behav Immun 2015;46:319Y326. doi:10.1016/ j.bbi.2015.02.023.
91. Sumowski JF, Rocca MA, Leavitt VM, et al. Searching for the neural basis of reserve against memory decline: intellectual enrichment linked to larger hippocampal volume in multiple sclerosis. Eur J Neurol 2015; doi:10.1111/ene.12662.
92. Zhu N, Jacobs DR Jr, Schreiner PJ, et al. Cardiorespiratory fitness and brain volume and white matter integrity: the CARDIA Study. Neurology 2015;84(23):2347Y2353. doi:10.1212/WNL.0000000000001658.
93. Tettey P, Simpson S Jr, Taylor BV, van der Mei IA. Vascular comorbidities in the onset and progression of multiple sclerosis. J Neurol Sci 2014;347(1Y2):23Y33. doi:10.1016/ j.jns.2014.10.020. 94. Tettey P, Simpson S Jr, Taylor B, et al. An adverse lipid profile is associated with disability and progression in disability, in people with MS. Mult Scler 2014;20(13): 1737Y1744. doi:10.1177/1352458514533162.
