Relationships Among Physical Inactivity Deconditioning, and Walking Impairment in Persons With Multiple Sclerosis
Why is this important to me?
Problems with walking are common if you have MS and can affect your ability to perform daily activities and remain employed as well as your quality of life. The MS disease process may lead you to be physically inactive, which may cause you to be out of shape, leading to worsening of MS and problems such as walking impairment. People with MS often engage in less vigorous physical activity, are less physically fit, and have impaired walking abilities compared to healthy individuals. The relationship among the three factors, physical activity, physical fitness, and walking ability, is not clear.
What is the objective of this study?
The authors compared 31 individuals with MS and 31 people without a chronic illness. In each participant, the authors measured:
- Moderately vigorous physical activity from a device worn by the participants that measured the amount of time the person performed vigorous physical activity over a 7-day period
- Physical fitness by measuring aerobic capacity when participants rode a stationary bicycle
- Walking ability by how far each participant could walk during 6 minutes
Results showed:
- The MS group (22 minutes per day) was less physically active than the non-MS group (31 minutes per day). Neither group met public health guidelines for physical activity.
- The MS group was less physically fit than the non-MS group.
- The MS group was able to walk a shorter distance in 6 minutes compared to the non-MS group.
- Time spent performing physical activity and physical fitness were associated with walking ability in the MS group, the non-MS group, and the two groups combined.
- Having MS partially explained differences in walking ability between people with and without MS.
- Physical activity and physical fitness partially explained differences in walking ability between people with and without MS.
Thus, direct effects of MS and indirect effects of MS that occurred via reduced physical activity and lower physical fitness contributed to reduced walking ability in people with MS.
These results suggest that being physically out of shape, perhaps due to physical inactivity, may lead to impaired walking ability if you have MS. This study was only able to show associations among factors, and did not show that one factor caused another. Additional studies will be needed for such conclusions. Nevertheless, if you have MS, increasing your physical activity may improve your physical fitness, which may in turn increase your walking ability.
This study did not measure other factors that may impact walking ability in people with MS such as overall disability, fatigue, and brain lesions. In addition, because this study only enrolled people with MS with mild disability, the results of this paper may or may not apply to more severely disabled people with MS.
How did the authors study this issue?
The authors enrolled 31 people with MS who had only mild disability due to MS and who could walk (with or without an assistive device). The authors also enrolled 31 non-MS people who were matched to the MS participants according to age, sex, height, and weight. Each person wore an accelerometer on his or her waist (at all time for 7 days except when bathing or swimming), which measured how much time the person spent performing vigorous physical activity. Participants exercised on a stationary bicycle, and their aerobic capacity was measured to determine their physical fitness. Each person also was asked to walk up and down a hallway with no obstacles for 6 minutes, and the distance walked was measured. People with MS were allowed to use an assistive device for walking if needed. The authors performed statistical analysis to understand the relationship among having or not having MS, physical activity, physical fitness, and walking ability. Each group contained 27 females and four males, with an average age of 42 (non-MS group) or 43 (MS group) years.
| SHARE: | |||||
Original Article
Relationships Among Physical Inactivity, Deconditioning, and Walking Impairment in Persons With Multiple Sclerosis
Journal of Neurologic Physical Therapy
Brian M. Sandroff, MS, Rachel E. Klaren, BS, and Robert W. Motl, PhD
Background and Purpose: We have previously proposed a conceptual model of physical inactivity, physiological deconditioning, and walking impairment in persons with multiple sclerosis (MS) that suggests engaging in physical activity and improving physiological conditioning may lead to improvements in walking performance. This cross-sectional study examined the nature of associations among physical activity, aerobic capacity, and walking performance in persons with MS and healthy controls.
Methods: The sample included 31 persons with MS and 31 controls matched by age, sex, height, and weight. Participants completed the 6-minute walk (6MW), wore an ActiGraph model GT3X accelerometer for 7 days as an objective measure of physical activity (expressed as time spent in moderate-to-vigorous physical activity [MVPA]), and completed an incremental exercise test on a cycle ergometer as a measure of aerobic capacity (VO2peak).
Results: Hierarchical linear regression analyses indicated that MVPA and VO2peak partially explained group differences in 6MW performance (R2 = 0.782), although group remained a significant predictor of 6MW performance (β = 0.304; P < 0.001). Path analysis indicated that group had both statistically significant direct and indirect effects on 6MW performance, and the indirect effect operated through pathways involving MVPA and VO2peak.
Discussion and Conclusions: These results provide direct preliminary evidence that physiological deconditioning, perhaps occurring as a result of physical inactivity, may explain variability in walking impairment in persons with MS. These findings support the design and implementation of targeted interventions for improving walking impairment in this population. Video Abstract available for more insights from the authors (see Supplemental Digital Content 1, http://links.lww.com/JNPT/A93).
INTRODUCTION
Multiple sclerosis (MS) is a common1 immune-mediated, multifocal demyelinating disease of the central nervous system2 that results in a cycle of physical inactivity, deconditioning, and walking impairment.3,4 Indeed, there is evidence that persons with MS engage in less moderate-to-vigorous physical activity5,6 (MVPA; ie, on the basis of rates of accelerometer output7), have lower peak aerobic capacity (ie, VO2peak; cardiorespiratory fitness),8,9 and have lower walking endurance performance (ie, 6-minute walk [6MW] distance)10,11 compared with healthy controls. There is further evidence that physical inactivity (ie, reduced MVPA) and physiological deconditioning (ie, compromised or reduced aerobic capacity) are both associated with walking impairments in persons with MS.12-15 Accordingly, we have developed the conceptual model that was based, in part, on empirical evidence describing the cross-sectional associations among physical inactivity and physiological deconditioning with walking impairment in MS12-17 (Figure 1). This model hypothesizes that the MS disease process results in physical inactivity that, in turn, initiates physiological deconditioning and ultimately worsening of MS, as indicated by functional manifestations (eg, walking impairment).9 The worsening of MS results in further physical inactivity and subsequent physiological deconditioning, yielding a cycle of associations among physical inactivity, deconditioning, and continual worsening of MS that develops over time. This model is of value as it conceptualizes the promotion of physical activity as a lifestyle behavior for maintaining and improving physical fitness levels and functional status in persons with MS.3 Parts of this model have been supported by results from different cross-sectional studies; however, the constructs have not previously been explicitly and simultaneously evaluated to examine whether physical inactivity, by way of physiological deconditioning, is associated with walking impairment in ambulatory persons with MS. To adequately test the tenets of the model, it is important to first establish whether MVPA and VO2peak can explain group differences (ie, persons with MS vs healthy controls) in walking performance using a regression-based approach, and then directly test the model (ie, whether MVPA and VO2peak are mediators of the association between MS and walking performance in ambulatory persons with MS) using path analysis.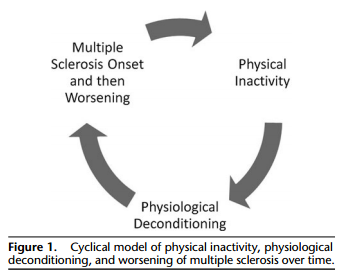
To that end, the current study examined the nature of associations among MVPA, aerobic capacity, and walking performance in ambulatory persons with MS and age, sex, height, and weight-matched controls. The primary aim of this study was to quantify the relationships among inactivity, deconditioning, and walking impairment9 to test the hypothesis that physical activity, by way of aerobic capacity, explains differences in 6MW performance among ambulatory persons with MS and controls. If such a hypothesis is supported, these data would provide preliminary evidence for the design and delivery of physical activity and exercise training interventions for managing walking impairment in ambulatory persons with MS by improving aerobic fitness. 3
METHODS
Participants
The current study represents a secondary analysis of data on physical inactivity, physiological deconditioning, and walking impairment; the original dataset was part of an investigation of physical fitness and cognition in persons with MS.18 The sample for both the original and current datasets included 31 persons with MS along, with 31 healthy controls matched by age, sex, height, and weight. The MS subsample was recruited prospectively from a preexisting database consisting of participants from previous studies conducted in our laboratory who provided a physician’s confirmation of MS and its clinical course.19-21 Healthy controls were prospectively recruited via public e-mail postings describing the study as an examination of physical fitness and cognition. The inclusion criteria for those with MS included (a) being ambulatory with or without an assistive device; (b) relapse free for the past 30 days; (c) 18 to 54 years of age; (d) willing and able to complete the physical activity, fitness, and walking assessments on the basis of self-report; and (e) having a low safety risk for maximal exercise testing on the basis of a “no” response to all items on the Physical Activity Readiness Questionnaire22 or a single “yes” response along with a physician’s approval. The same inclusion criteria were applied for the controls, with the exception of being relapse free for the past 30 days, and each control matched a person with MS on age, sex, height, and weight. Overall, 63 persons with MS were contacted and 51 expressed interest in participating. Of the 51 persons, 47 underwent screening over the telephone and 6 did not meet inclusion criteria; 10 persons cancelled the testing session (for reasons unrelated to testing). This resulted in a subsample of 31 persons with MS. We contacted 101 healthy controls, with 61 expressing interest. Of the 61 who were interested in participation, 32 were matched by age, sex, height, and weight with persons with MS, and 1-matched control did not meet inclusion criteria, resulting in a final subsample of 31 matched controls.
Protocol
The current study was approved by University of Illinois at Urbana-Champaign University Institutional Review Board, and all participants initially provided written informed consent. Regarding MVPA measurement, participants were given an accelerometer (ActiGraph model GT3X, Health One Technology, Fort Walton Beach, FL) to wear on an elastic belt around the waist on the side of the body in-line with the nondominant hip during the waking hours, except while showering, bathing, and swimming, over a 7-day period. In addition, participants were instructed to go about normal daily routines while wearing the device and not purposefully engage in additional physical activity or exercise behavior. Participants did not upload the accelerometer data; rather, the data were downloaded and processed by laboratory personnel after the receipt of the accelerometer from the participant. For measurement of aerobic capacity, participants underwent an incremental exercise test on a cycle ergometer to measure VO2peak. For the 6MW protocol, participants completed the 6MW in a hallway free of debris as per previous recommendations.10 Participants also completed a short battery of questionnaires that included a demographics form and Patient-Determined Disease Steps (PDDS). Upon completion of all procedures, participants were remunerated $30.
Measures
Physical Activity
Physical activity was objectively measured as the average number of minutes per day spent in MVPA using the accelerometers described in the previous section. The accelerometers were small (3.8 × 3.7 × 1.8 cm), lightweight (27 g), and contained a solid-state, digital accelerometer that generates an electrical signal proportional to the force acting on it. The accelerometer signal is digitized by a 12-bit, analog-to-digital converter at a rate of 30 Hz and integrated over a preprogrammed epoch interval; the epoch was 1 minute in this study. This epoch is consistent with previous research on free-living physical activity in large samples of persons with MS6 and persons in the general population.23 The integrated value is stored in memory as activity counts per minute, and the integrator is reset after each epoch interval. The data were retrieved from the accelerometer via a direct USB 2.0 connection with a personal computer and then imported into the associated software system (ActiLife 5) for validity check (ie, ≥10 hours wear time without periods exceeding 60 minutes of continuous zeroes per day). Days with less than 600 minutes of accelerometer wear time (ie, invalid day) were not included in the analyses, and only cases with at least 3 valid days of wear time were included in subsequent analyses. This approach is considered acceptable for generating a reliable estimate of usual physical activity in persons with MS.24 The primary outcome from the accelerometer was average minutes spent in MVPA per valid day. Processing of average minutes spent in MVPA per day using established cut points of 1584, and 1950 counts per minute for persons with MS and controls, respectively,7 was further completed by the processing software. Those cut points were generated in a previous calibration trial on the basis of associations between accelerometer output and energy expenditure (ie, oxygen consumption) during various speeds of treadmill walking in persons with MS and healthy controls matched by age, sex, height, and weight.7 The strength and slope of those associations differed between persons with MS and matched controls; persons with MS had similar rates of activity counts, but expended significantly more energy than matched controls across a range of speeds. This resulted in the development of the above-noted cut points for MVPA.7 Those cut points are valid in persons with MS5 and average minutes of MVPA per day is a useful expression for comparing rates of physical activity with public health guidelines.6 We did not examine sedentary minutes or minutes spent in light physical activity on the basis of the accelerometer data, as time spent in MVPA is the currently accepted metric for public health guidelines for physical activity,25 and empirically derived cut points for sedentary and light physical activity for persons with MS have not yet been established. We do not believe that the same cut points for the general population of adults would be applicable for persons with MS on the basis of the difference observed for the cut points for MVPA in MS versus adults without MS.7 Throughout the article, we refer to the primary physical activity outcome as “MVPA” and thereby refer to this as “physical inactivity” (i.e., low levels of MVPA) when situating this variable within the context of the previously hypothesized model of inactivity, deconditioning, and walking impairment in persons with MS.9
Aerobic Capacity
Aerobic capacity was measured as peak oxygen consumption (VO2peak) using an incremental exercise test on an electronically braked, computer-driven cycle ergometer (Lode BV, Groningen, the Netherlands) and a calibrated open-circuit spirometry system (TrueOne, Parvo Medics, Sandy, UT) for analyzing expired gases. As previously reported,15,18 the incremental exercise test was preceded by a 3-minute warmup at 0 W. The initial work rate for the incremental exercise test was 0 W, and the work rate continuously increased at a rate of 15 W/min until the participant reached volitional fatigue (ie, inability to continue exercising). This protocol has been validated in persons with MS for measuring peak aerobic capacity.8 Oxygen consumption (VO2), respiratory exchange ratio, and work rate were measured continuously by the open circuit spirometry system and expressed as 20-second averages. Heart rate (HR) was displayed using a heart rate monitor (Polar Electro Oy, Finland), and heart rate and rate of perceived exertion (RPE) were recorded every minute. VO2peak was expressed in mL·kg−1·min−1 on the basis of the highest-recorded 20-second VO2 value when 2 of the 3 criteria were satisfied: (1) respiratory exchange ratio of 1.10 or more; (2) peak heart rate within 10 beats per minute of age-predicted maximum (ie, ∼1 standard deviation [SD]); or (3) peak RPE of 17 or more. Throughout this report, we refer to aerobic capacity as “VO2peak” and thereby refer to this as “physiological deconditioning” (i.e., lower VO2peak) when situating this variable within the context of the previously hypothesized model of inactivity, deconditioning, and walking impairment in persons with MS.9
Walking Performance
The 6MW was administered as a measure of walking endurance and is a valid and reliable test of endurance walking performance in persons with MS.10,13,26 Although we recognize that the 6MW is primarily considered a test of walking endurance in this population, for the purposes of testing our hypothesis, we operationalized the 6MW as an indication of walking impairment in persons with MS. We based this decision on the strong correlations between the 6MW and other valid measures of walking performance (ρ’s > 0.80)26 and markedly strong correlation between 6MW and disability status as measured by the Expanded Disability Status Scale (EDSS) (r = −0.73)10 in persons with MS. We recognize that the 6MW is often used as a measure of aerobic capacity in other populations (eg, persons with heart failure)27; however, in persons with MS, there is evidence that 6MW performance may be more strongly associated with free-living walking behavior (ie, steps per day) than with measures of physical function.11 Therefore, we included 6MW performance in our model as a measure of walking endurance rather than VO2peak. As such, throughout the article, we refer to total distance traveled during the 6MW as “6MW performance” and thereby refer to this as “walking impairment” (i.e., poor 6MW performance) when situating this variable within the context of the previously proposed hypothesis of inactivity, deconditioning, and walking impairment.9 Regarding the 6MW test, participants were instructed to walk as fast and as far as possible for 6 minutes within a single corridor that was approximately 23 m (75 ft) in length and performing 180◦ turns, and, when necessary, participants were permitted to use an assistive device.28 The 6MW was administered using standardized instructions for persons with MS (ie, walking as fast and far as possible without rest or encouragement for 6 minutes).10 One researcher followed approximately 1 m (3 ft) behind the participant with a measuring wheel (Stanley MW50, New Briton, CT) and recorded total distance traveled. All participants were further able to complete the 6MW without incident.
Ambulatory Disability
The PDDS scale27 was used as a measure of self-reported ambulatory disability status. The PDDS was developed as an inexpensive self-report surrogate of the Expanded Disability EDSS and contains a single item for measuring self-reported neurological impairment on an ordinal level, ranging from 0 (normal) through 8 (bedridden). Scores from the PDDS are linearly and strongly related with EDSS scores (ρ = 0.78), 6MW distance (ρ = 0.81), Multiple Sclerosis Walking Scale- 12 scores (ρ = 0.80), and free-living steps per day from an accelerometer (ρ = −0.74).30
Data Analysis
Data were analyzed in SPSS version 21 (SPSS Inc, Chicago, IL). We first examined differences between MS and control groups in age, height, and weight using independent samples t tests. Independent samples t tests were then conducted to examine differences between MS and control groups in time spent in MVPA, VO2peak, and 6MW performance. The magnitude of differences between groups was expressed as Cohen’s d (ie, difference in mean scores between groups divided by the pooled SD)31; with values of 0.2, 0.5, and 0.8 interpreted as small, moderate, and large, respectively, on the basis of recommended boundary values for expressing the magnitudes of effect sizes.31 Bivariate Pearson product-moment correlations (r) were then performed between measures of physical activity, aerobic capacity, and 6MW performance in the overall sample (MS and control groups combined) and in the MS and healthy control groups, separately. Values for correlation coefficients of 0.1, 0.3, and 0.5 were interpreted as small, moderate, and large, respectively, on the basis of recommended boundary values for expressing the magnitudes of correlation coefficients.31
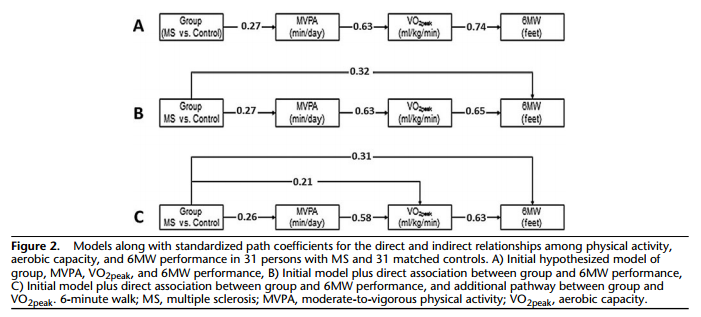
The next analysis involved hierarchical linear regression with direct entry to examine whether MVPA and VO2peak accounted for MS-related variance (ie, group differences; MS vs control; MS group coded as 1, control group coded as 2) in 6MW performance. This was undertaken by regressing 6MW performance on group in Step 1 and then adding time spent in MVPA in Step 2, and VO2peak in Step 3; this order of entry is based on our conceptual model. We compared the β-coefficients for group in Step 1, Step 2, and Step 3 to examine whether MVPA and VO2peak accounted for group differences in 6MW performance; we further examined the β-coefficients of MVPA and VO2peak in Step 2 and Step 3 to identify the independent contributions of those variables for explaining differences in 6MW performance. These were precursory steps before conducting the path analysis using Mplus (Muthen & Muthen, Los Angeles, CA)31 for directly testing the indirect pathway between group and 6MW by way of MVPA and VO2peak (Figure 2A). We evaluated model fit on the basis of a nonsignificant chi-square statistic, comparative fit index (CFI) more than 0.95, and standardized root mean square residual (SRMR) less than 0.06. We inspected modification indices in the event of poor model fit, and then adjusted and retested the model accordingly.
RESULTS
Participants Characteristics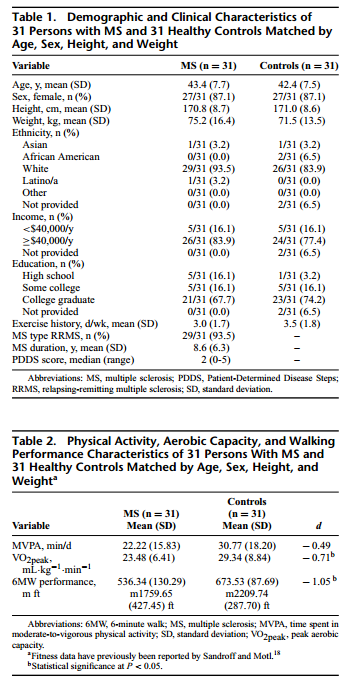
Participant characteristics for the 31 persons with MS and 31 matched controls are presented in Table 1. Briefly, there were no differences in age (t = 0.57; P = 0.57), height (t = −0.11; P = 0.91), or weight (t = 0.95; P = 0.35) between the groups, and the groups were matched evenly on sex (27 females and 4 males). Among those with MS, 29 participants (94%) had a relapsing-remitting disease course, and 2 participants (6%) had secondary-progressive MS; the mean duration of MS was 8.6 (SD = 6.3) years. The MS sample had further minimal ambulatory disability on the basis of PDDS scores (median = 2; range = 0-5); this was expected given the inclusion/exclusion criteria on the basis of the demanding nature of the fitness test.
MVPA, VO2peak, and 6MW Performance
The descriptive statistics for MVPA, VO2peak, and 6MW performance for the MS and control groups are presented in Table 2; the effect sizes for group differences in these measures are further provided in Table 2. There were 58 participants who provided sufficient accelerometer data. Four participants (2 MS, 2 healthy controls) provided insufficient accelerometer data (ie, under 3 valid days of wear time). The 58 cases with sufficient accelerometer data were included in subsequent analyses examining group differences in MVPA. All 62 participants completed the incremental exercise test on the cycle ergometer and the 6MW. Thus, all 62 cases were included in the analyses examining group differences in VO2peak and 6MW performance, respectively. The full sample size of 62 was included in the correlation analyses, as those involved pairwise comparisons. The full sample size of 62 was further included in the path analyses, as Mplus uses the full-information maximum likelihood estimator as default for accounting for missing data (ie, MVPA) (ie, the model is estimated in the presence of all available cases and data).
Participants with MS wore the accelerometer for an average of 840.9 minutes per day and 5.75 valid days over the 7-day period. There were no statistically significant differences in MVPA between persons with MS who wore the accelerometer for 7 valid days (n = 7) and those who wore the accelerometer for 3 to 6 valid days (P = 0.80). Healthy controls wore the accelerometer for an average of 856.9 minutes per day and 5.83 valid days over the 7-day period. There were no statistically significant differences in MVPA between healthy control participants who wore the accelerometer for 7 valid days (n = 9) and those who wore the accelerometer for 3 to 6 valid days (P = 0.73). of valid days per week and minutes per day were not significantly different between MS and healthy control groups on the basis of independent-samples t-tests (P > 0.41).
Eighty-seven percent of the MS subsample (ie, n = 27) and 94% of the healthy control subsample (ie, n = 29) met 2 of the 3 criteria for determining VO2peak; those differences between groups were not statistically significant (P = 0.39). Furthermore, VO2peak did not significantly differ between those who met at least 2 criteria and those who did not within the MS (P = 0.20) and control (P = 0.97) groups, respectively. Mean respiratory exchange ratio was 1.19 (SD = 0.06) and 1.23 (SD = 0.09) for persons with MS and healthy controls, respectively.
Group Differences in MVPA, VO2peak, and 6MW Performance
Regarding the primary outcome measures, the MS group engaged in 22.2 minutes per day of MVPA, which does not meet public health guidelines,25 demonstrated aerobic capacity levels that ranked below the 10th percentile among women aged 40 to 49 years,32 and substantially shorter 6MW distances than other samples of healthy controls.10 By comparison, the healthy control group engaged in 30.8 minutes per day of MVPA, which does meet public health guidelines,25 demonstrated aerobic capacity levels that ranked above the 30th percentile among women aged 40 to 49 years,32 and similar 6MW distances as other samples of healthy adults.10 There were significant differences in time spent in MVPA (t = −1.91; P = 0.031; d =−0.49), VO2peak (t =−2.99; P = 0.004; d =−0.71), and 6MW performance (t = −4.86; P < 0.001; d = −1.05), on the basis of 1-tailed tests given our directional hypotheses, between persons with MS and matched controls. The effect sizes of −0.49, −0.71, and −1.05 indicated that persons with MS spent moderately less time in MVPA, had moderately reduced VO2peak, and substantially shorter 6MW distance compared with matched controls, respectively. Collectively, this suggests that persons with MS demonstrated physical inactivity, physiological deconditioning, and walking impairment relative to matched controls.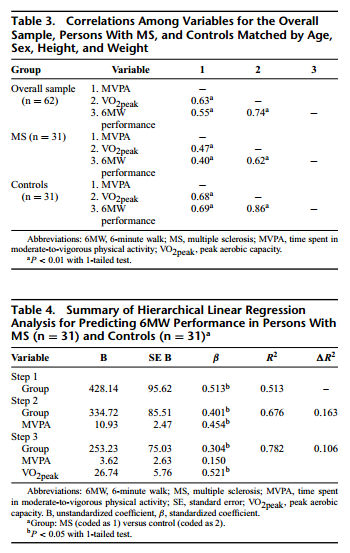
Correlation Analysis
Bivariate correlations among MVPA, VO2peak, and 6MW performance for the overall, MS, and control groups are reported in Table 3. Time spent in MVPA and aerobic capacity were associated with 6MW performance in the overall, MS, and control groups, and these correlations were moderate to large in magnitude (r = 0.40-0.86). The correlation between aerobic capacity and 6MW performance in MS indicates that these variables are measuring the same construct (ie, 38.4% shared variance). Time spent in MVPA was further associated with VO2peak in the combined, MS, and control groups, and these correlations were moderate to large in magnitude (r = 0.47-0.68). This indicates that physical inactivity, physiological deconditioning, and walking impairment were intercorrelated in persons with MS.
Regression Analysis
The results for the hierarchical regression analysis are shown in Table 4. Group (B = 428.14; standard error (SE) B = 95.62; β = 0.513) explained a statistically significant (F(1, 57) =20.047; P<0.001) portion of variance in 6MW performance (R2 = 0.513) in Step 1. This indicates that having MS explained a significant portion of variance in walking performance. In Step 2 of the model, the β-coefficient for group became attenuated (β = 0.401; P < 0.001) and, along with time spent in MVPA (B = 10.93; SE B = 2.47; β = 0.454), explained a statistically significant amount of variance in 6MW performance (R2 = 0.676). This indicates that MVPA partially explained MS-related variance in walking performance. In Step 3 of the model, the β-coefficient for group further became attenuated (β = 0.304; P < 0.001) and along with VO2peak (B = 26.74, SE B = 5.76, β = 0.521), explained a statistically significant (F(3, 54) = 28.387, P < 0.001) amount of variance in 6MW performance (R2 = 0.782); the β-coefficient for MVPA became attenuated and nonsignificant (β = 0.150; P = 0.175). This indicates that MVPA and VO2peak partially accounted for MSrelated variance in 6MW performance among persons with MS and controls.
Path Analysis
We tested the proposed and modified models in Figure 2 using path analysis; we provide standardized path coefficients for the direct and indirect relationships among MVPA, VO2peak, and 6MW performance in text and in the figure. The standardized path coefficients are interpreted such that an incremental unit change in one variable is associated with an incremental SD unit change in a second variable. We originally tested the proposed model that group (ie, MS versus control) was indirectly associated with 6MW performance through a pathway that involved both MVPA and VO2peak (Figure 2A). This model had poor fit (χ2(df = 6) = 94.81; P < 0.01; CFI = 0.78; SRMR = 0.12). On the basis of modification indices, we then tested a second model that further included a direct association between group and 6MW performance (Figure 2B). The second model had improved, but not good fit (χ2(df = 2) = 9.50; P = 0.01; CFI = 0.92; SRMR = 0.08), and this improvement was based on a chi-square difference test (P < 0.05) and comparison of the CFI and SRMR between models. On the basis of modification indices, we tested a third model (Figure 2C) that included an additional pathway between group and VO2peak. The third model had good fit (χ2(df = 1) = 3.66; P = 0.06; CFI = 0.97; SRMR = 0.02), and fit better than model 2 again on the basis of a chi-square difference test (P < 0.05) and comparison of the CFI and SRMR between models. The third model indicated that group had statistically significant direct and indirect effects on 6MW performance. The indirect effects occurred through pathways that involved only VO2peak (standardized path coefficient = 0.13) or both MVPA and VO2peak (standardized path coefficient = 0.10), but the direct effect of group on 6MW performance was still statistically significant (standardized path coefficient = 0.31).
DISCUSSION
This cross-sectional study is the first to directly examine the nature of associations among MVPA, VO2peak, and 6MW performance in ambulatory persons with MS and healthy controls matched by age, sex, height, and weight. The primary findings indicated that (a) there were significant differences in MVPA, VO2peak, and 6MW performance between persons with MS and matched controls; (b) MVPA and VO2peak were associated with 6MW performance in the overall sample, persons with MS, and matched controls; (c) MVPA and VO2peak partially accounted for group differences in 6MW performance; and (d) group had both statistically significant direct and indirect effects through pathways involving MVPA and VO2peak on 6MW performance. Collectively, these results provide direct preliminary evidence that physiological deconditioning, perhaps occurring as a result of physical inactivity, may explain variability in walking impairment in ambulatory persons with MS.3,9 Importantly, this provides an empirical basis for informing researchers and clinicians of potential targets (ie, increasing physical activity and improving aerobic capacity) for improving walking function among ambulatory persons with MS.
Walking impairment is a highly prevalent, disabling, and poorly managed consequence of the MS disease process33-35 and is associated with limitations in performing activities of daily living, unemployment, and compromised quality of life.34,35 We have previously proposed a model of physical inactivity, physiological deconditioning, and walking impairment in persons with MS that suggests engaging in physical activity and improving physiological conditioning may lead to improvements in walking performance.3 The current study directly tested the tenets of that hypothesis by examining differences in and associations among time spent in MVPA, VO2peak, and 6MW performance in persons with MS and a comparison sample of matched controls. The results support the hypothesis that physical inactivity (ie, MVPA) and physiological deconditioning (ie, VO2peak) partially explain group differences in walking impairment (ie, 6MW performance) between persons with MS and controls.9 The current results from the path analysis provide further support for this hypothesis 41−43 that group (ie, MS vs control) was directly associated with 6MW performance, and indirectly associated with 6MW performance by way of MVPA and VO2peak. Collectively, these results favor the design and implementation of longitudinal interventions for improving walking performance in ambulatory persons with MS by increasing lifestyle physical activity (ie, daily accumulation of at least 30 minutes of self-selected activities that are at least of moderate-to-vigorous intensity that could be planned or unplanned, that occur in everyday life36) through a behavioral intervention and/or improving physiological conditioning (ie, VO2peak) through exercise training.
Behavioral interventions involve systematic instruction on skills and techniques for modifying health-related behaviors including physical activity; such behavioral interventions have been successful for increasing physical activity behavior in persons with MS.37 Alternatively, exercise training involves maintaining and improving physical fitness levels and has been previously identified as an approach to improve mobility in this population.9,38,39 The current results suggest that one approach may involve first administering behavioral interventions for increasing MVPA, which may result in improved physiological conditioning, and in turn, improvements in ambulation (ie, secondary prevention) in persons with relatively mild MS disability. However, the comparative or additive effects of physical activity and exercise training on walking performance are unknown in ambulatory persons with MS, and should be subject to future examination. We acknowledge that this study involved persons with relatively mild ambulatory disability, and that there are limitations in increasing physical activity and improving physical fitness in persons with advanced MS that will need to be overcome in future research.4,40
One unique aspect of the current study was that we directly examined MVPA and VO2peak as mediator variables of the relationship between MS and 6MW performance. Results from the preliminary hierarchical linear regression analysis indicated that even after accounting for MVPA and VO2peak, group (ie, MS vs control) still significantly explained differences in 6MW performance (β = 0.304; P < 0.001). This suggests that physical inactivity and physiological deconditioning were partial mediators of the association between MS and walking impairment. Results from the path analysis con- firmed that group had a statistically significant direct effect on 6MW performance, and statistically significant indirect effects through pathways involving MVPA and VO2peak (ie, partial mediation; Figure 2C). On the basis of these results, it is important to note that other factors (eg, global disability, symptoms, fatigue, body composition, and lesion load volume) may substantially contribute to walking impairment apart from physical inactivity and physiological deconditioning41-43 and should be taken into account in future longitudinal investigations. Strengths of the current study include the objective measurement of MVPA, VO2peak, and 6MW performance; inclusion of healthy controls matched by age, sex, height, and weight; and direct examination of the constructs within a previously hypothesized model.
Limitations
The primary limitation of this study is the cross-sectional nature of the investigation, as it does not indicate causality and temporal patterns of the association between physical inactivity, physiological deconditioning, and walking impairment. Nevertheless, we provide preliminary data necessary before investing considerable time, effort, and resources into a longitudinal intervention on physical activity, physical fitness, exercise training, and walking performance in persons with MS. Further limitations of the current study include the relatively small sample size (ie, N = 62), and relatively mild ambulatory disability status of persons with MS (ie, median PDDS score = 2), respectively. The small sample size and inclusion of a sample with mild disability was necessary given the nature of the outcomes, but limits the generalizability of our results broadly among persons with MS. Finally, the current study did not include a global measure of disability (ie, EDSS), measures of symptoms, fatigue, other measures of fitness (eg, body composition), and brain structure/function as other potential mediator variables of the association between MS and walking impairment.
CONCLUSIONS
This study examined MVPA, VO2peak, and 6MW performance in a secondary analysis of a cross-sectional study of physical fitness and cognition in persons with MS and controls.18 There were significant differences in MVPA, VO2peak, and 6MW performance between persons with MS and matched controls; MVPA and VO2peak were associated with 6MW performance in the overall sample, persons with MS, and matched controls; MVPA and VO2peak partially accounted for group differences in 6MW performance; and group had a statistically significant direct and indirect effects (ie, through pathways involving MVPA and VO2peak) on 6MW performance. Collectively, these results provide direct support for the hypothesis that physiological deconditioning, perhaps because of physical inactivity, may explain variability in walking impairment in persons with MS.3 The current study provides additional rationale for the design and implementation of physical activity and exercise training interventions as approaches for improving walking performance in this population.
REFERENCES
1. Mayr WT, Pittock SJ, McClelland RL, Jorgensen NW, Noseworthy JH, Rodriguez M. Incidence and prevalence of multiple sclerosis in Olmstead County, Minnesota, 1985-2000. Neurology. 2003;61:1373-1377
2. Trapp BD, Nave K. Multiple sclerosis: an immune or neurodegenerative disorder? Annu Rev Neurosci. 2008;31:247-269.
3. Motl RW. Physical activity and irreversible disability in multiple sclerosis. Exerc Sport Sci Rev. 2010;38(4):186-191.
4. Motl RW, Pilutti LA, Sandroff BM. The importance of physical fitness in multiple sclerosis. J Nov Physiother. 2013;3(2):141-147.
5. Sandroff BM, Dlugonski D, Weikert M, Suh Y, Balantrapu S, Motl RW. Physical activity and multiple sclerosis: new insights regarding inactivity. Acta Neurol Scand. 2012;126(4):256-262.
6. Klaren RE, Motl RW, Dlugonski D, Sandroff BM, Pilutti LA. Objectively quantified physical activity in persons with multiple sclerosis. Arch Phys Med Rehabil. 2013;94(12):2342-2348.
7. Sandroff BM, Motl RW, Suh Y. Accelerometer output and its association with energy expenditure in persons with multiple sclerosis. J Rehabil Res Dev. 2012;49(3):467-476.
8. Motl RW, Fernhall B. Accurate prediction of cardiorespiratory fitness using cycle ergometry in minimally disabled persons with relapsing-remitting multiple sclerosis. Arch Phys Med Rehabil. 2012;93(3):490-495.
9. Motl RW, Goldman M. Physical inactivity, neurological disability, and cardiorespiratory fitness in multiple sclerosis. Acta Neurol Scand. 2011;123(2):98-104.
10. Goldman MD, Marrie RA, Cohen JA. Evaluation of the six-minute walk in multiple sclerosis subjects and healthy controls. Mult Scler. 2008;14(3):383-390.
11. Motl R, Suh Y, Balantrapu S, et al. Evidence for the different physiological significance of the 6- and 2-minute walk tests in multiple sclerosis. BMC Neurol. 2012;12(1):6.
12. Gijbels D, Alders G, Van Hoof E, et al. Predicting habitual walking performance in multiple sclerosis: relevance of capacity and self-report measures. Mult Scler. 2010;16(5):618-626.
13. Motl RW, Sandroff BM, Suh Y, Sosnoff JJ. Energy cost of walking and its association with gait parameters, daily activity, and fatigue in persons with mild multiple sclerosis. Neurorehabil Neural Repair. 2012;26(8):1015- 1021.
14. Motl RW, Pilutti L, Sandroff BM, Dlugonski D, Sosnoff JJ, Pula JH. Accelerometry as walking behavior in multiple sclerosis. Acta Neurol Scand. 2013;127(6):384-390.
15. Sandroff BM, Sosnoff JJ, Motl RW. Physical fitness, walking performance, and gait in multiple sclerosis. J Neurol Sci. 2013;328(1-2):70-76.
16. Thoumie P, Lamotte D, Cantalloube S, Faucher M, Amarenco G. Motor determinants of gait in 100 ambulatory patients with multiple sclerosis. Mult Scler. 2005;11(4):485-491.
17. Motl RW, Goldman MD, Benedict RH. Walking impairment in patients with multiple sclerosis: exercise training as a treatment option. Neuropsychiatr Dis Treat. 2010;16:767-774.
18. Sandroff BM, Motl RW. Fitness and cognitive processing speed in persons with multiple sclerosis: a cross-sectional investigation. J Clin Exp Neuropsychol. 2012;34(10):1041-1052.
19. Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology. 1996;46(4):907-911.
20. McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the Diagnosis of Multiple Sclerosis. Ann Neurol. 2001;50(1): 121-127.
21. Poser CM, Paty DW, Scheinberg L, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol. 1983;13(3): 227-231.
22. Thomas S, Reading J, Shephard RJ. Revision of the Physical Activity Readiness Questionnaire (PAR-Q). Can J Sport Sci. 1992;17(4): 338-345.
23. Troiano RP, Berrigan D, Dodd KW, Masse LC, Tilert T, McDowell M. Physical activity in the United States measured by accelerometer. Med Sci Sports Exerc. 2008;40:181-8.
24. Motl RW, Zhu W, Park Y, McAuley E, Scott JA, Snook EM. Reliability of scores from physical activity monitors in adults with multiple sclerosis. Adapt Phys Activ Q. 2007;24(3):245-253.
25. Garber CE, Blissmer B, Deschenes MR, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43:1334-1359.
26. Kieseier BC, Pozzilli C. Assessing walking disability in multiple sclerosis. Mult Scler. 2012;18(7):914-924.
27. Cahalin LP, Mathier MA, Semigran MJ, Dec GW, DiSalvo DG. The six-minute walk test predicts peak oxygen uptake and survival in patients with advanced heart failure. Chest. 1996;110:325-332.
28. Sandroff BM, Pilutti LA, Dlugosnki D, et al. Comparing two conditions of administering the six-minute walk in persons with multiple sclerosis. Int J MS Care. 2014;16(1):48-54.
29. Hadjimichael O, Kerns RD, Rizzo MA, Cutter G, Vollmer T. Persistent pain and uncomfortable sensations in persons with multiple sclerosis. Pain. 2007;127(1-2):35-41.
30. Learmonth YC, Motl RW, Sandroff BM, Pula JH, Cadavid D. Validation of patient determined disease steps (PDDS) scale scores in persons with multiple sclerosis. BMC Neurol. 2013;13(1):37.
31. Cohen J. Statistical power analysis for the behavioral sciences. 2nd ed. Hillsdale, NJ: Lawrence Erlbaum Associates; 1988.
32. American College of Sports Medicine’s guidelines for exercise testing and prediction, seventh edition. Philadelphia, PA: Lippincott Williams & Wilkins; 2006.
33. Heesen C, Romberg A, Gold S, Schulz KH. Physical exercise in multiple sclerosis: supportive care or a putative disease-modifying treatment. Expert Rev Neurother. 2006;6:347-355.
34. Larocca NG. Impact of walking impairment in multiple sclerosis: perspectives of patients and care partners. Patient. 2011;4(3):189- 201.
35. Panitch H, Applebee A. Treatment of walking impairment in multiple sclerosis: an unmet need for a disease-specific disability. Expert Opin Pharmacother. 2011;12(10):1511-1521.
36. Dunn AL, Andersen RE, Jakicic JM. Lifestyle physical activity interventions. History, short- and long-term effects, and recommendations. Am J Prev Med. 1998;15:398-412.
37. Motl RW, Dlugonski D, Wojcicki TR, McAuley E, Mohr DC. Internet intervention for increasing physical activity in persons with multiple sclerosis. Mult Scler. 2011;17(1):116-128.
38. Rietberg MB, Brooks D, Uitdehaag BMJ, Kwakkel G. Exercise therapy for multiple sclerosis. Cochrane Database Syst Rev. 2004;3.
39. Garrett M, Coote S. Multiple sclerosis and exercise in people with minimal gait impairment - a review. Phys Ther Rev. 2009;14(3):169- 180.
40. Pilutti LA. Adapted exercise interventions for persons with progressive multiple sclerosis. Appl Physiol Nutr Metab. 2013;38(3):357.
41. Pilutti LA, Dlugonski D, Pula JH, Motl RW. Weight status in persons with multiple sclerosis: implications for mobility outcomes. J Obes. 2012;2012:868256. doi:10.1155/2012/868256.
42. Fisniku LK, Brex PA, Altmann DR, et al. Disability and T2 MRI lesions: a 20-year follow-up of patients with relapse onset of multiple sclerosis. Brain. 2012;131(Pt 3):808-817.
43. Motl RW, Balantrapu S, Pilutti L, et al. Symptomatic correlates of six-minute walk performance in persons with multiple sclerosis. Eur J Phys Rehabil Med. 2013;49(1):59-66.

