Physical Therapy Interventions and Urinary Incontinence
Why is this important to me?
Urinary incontinence (UI) affects many people with MS and leads to a decrease in quality of life. Strategies are needed to treat UI, but little information is available about the effectiveness of the various strategies including non-invasive treatments such as physical therapy.
Who will benefit from reading this study/article?
This article reviewed six previously published studies looking at the effectiveness of various types of physical therapy to treat UI and improve quality of life in men and women with MS.
What is the objective of this study?
UI affects more than 12% of the general population and 50-100% of people with MS. Physical therapy is effective for treating UI in the general population, but its effectiveness in people with MS is unclear.
UI includes “stress incontinence” and “urge incontinence”. Stress incontinence is when the pelvic floor muscles are too weak and urine leaks when a person laughs, coughs, or sneezes. Urge incontinence, which is also called over-active bladder, is when urine leaks right before a powerful urge to urinate.
UI is embarrassing and affects a person’s quality of life due to reduced physical and social function, decreased emotional health, limited participation in various activities, depression, and low self-esteem. Evidence for the effectiveness of various strategies for treating UI in people with MS is needed before these interventions can be widely encouraged. Interventions should be safe, well-tolerated, long-lasting, and preferably non-surgical.
UI can be managed conservatively or surgically. Various interventions may include:
- Self-catheterization
- Drug treatment
- Complementary and alternative medicine
- Minimally invasive surgery
- Pelvic floor muscle training (Kegel exercises), a type of physical therapy
- Neuromuscular electrical stimulation, a type of physical therapy that teaches the patient to control voluntary muscle movement through visual and auditory feedback
Electromyography biofeedback, a type of physical therapy that teaches the patient to control voluntary muscle movement through visual and auditory feedback
The authors reviewed six published studies to look for evidence that physical therapy improves quality of life and UI in people with MS.
The first study tested the effects of pelvic floor muscle training in 27 females with MS. The study showed improvement in UI and quality of life in those receiving the therapy compared to a group that did not receive therapy.
The second study looked at 24 females with MS who participated in a personalized physical and occupational therapy program compared to 34 females with MS who received no therapy. Quality of life improved and the amount of bother due to UI was reduced in the therapy group compared to the non-therapy group.
The third study examined the effect of pelvic floor exercises and electrical stimulation in 50 females and 30 males with MS. Compared to the group that received no therapy, the therapy group showed improvement in urgency, frequency, and UI. Males responded better than females.
In the fourth study, 30 females with MS were assigned to one of three treatments: pelvic floor muscle treatment advice; pelvic floor muscle treatment advice and electromyography biofeedback; or pelvic floor muscle treatment advice, electromyography biofeedback, and neuromuscular electrical stimulation. The combination of all three interventions improved UI in these women with MS.
The fifth study looked at 26 women and 11 men with MS who received pelvic floor muscle training, electromyography biofeedback, and placebo neuromuscular electrical stimulation and 31 women and 6 men with MS who received pelvic floor muscle training, electromyography biofeedback, and active neuromuscular electrical stimulation. Although separate results for women and men were not reported, the authors concluded that the combination of all three interventions alleviates some symptoms of UI in people with MS.
The sixth study involved non-invasive nerve stimulation with surface electrodes. Fifty-one women and 19 men with MS experience reduced urinary urgency and improved quality of life compared to before the study began.
Overall, a direct effect of these therapies on UI was seen. Effects of therapy on quality of life may be indirect due to influences on fatigue, muscle weakness, time needed to perform activities of daily living, and depression.
Physical therapy, especially pelvic floor muscle training, electrical stimulation, and biofeedback, improved quality of life and UI symptoms, and the interventions were safe and well tolerated. However, these studies have some limitations. The possibility of long-term maintenance of improvement is not known, and longer studies are needed. The physical therapy interventions varied widely, and which intervention or combination of therapies is best is not known. MS is unpredictable and involves several disease subtypes. Thus, a standard treatment plan for everyone with MS will be difficult to develop. A customized, conservative treatment plan may be needed for each person.
How did the authors study this issue?
The authors reviewed six previously published studies that used various non-invasive physical therapy interventions in people with MS who had UI. The effect of the intervention was assessed with questionnaires and objective measurements of urinary control.
| SHARE: | |||||
Original Article
Do Physical Therapy Interventions Affect Urinary Incontinence and Quality of Life in People with Multiple Sclerosis? An Evidence-Based Review
International Journal of MS Care
Valerie Block, PT; Monica Rivera, DPTSc; Marsha Melnick, PhD; Diane D. Allen, PhD
Multiple sclerosis (MS) is a chronic, degenerative disease of the central nervous system characterized by deterioration of the myelin and associated neuronal axons.1 The symptoms vary depending on the location of the lesion in the central nervous system and disease progression type. Common impairments include fatigue, ataxia, tremor, spasticity, bladder or bowel dysfunction, impaired vision, pain, cognitive disorders, dysphagia, and sexual dysfunction.1,2 MS has no clear cause and, as yet, no definite cure.3,4
Urinary incontinence (UI), one of the common dysfunctions in MS, is also prevalent in the general population. In the United States, more than 12.6% of the general population has been diagnosed as having incontinence, with the worldwide prevalence estimated to be 115 million in 2013.5–7 In contrast to the non-MS population, the prevalence of UI in people with MS is 50% to 100%.8,9 If someone with MS also has vision and balance impairments, having UI poses important health and safety concerns, with an increase in fall risk while rushing to the restroom.2,3,9,10
Physical therapy (PT) has proved to be effective in reducing the effects of UI in non-MS populations, but literature is scarce in MS.11,12 The purpose of this study was to review the existing literature regarding the effectiveness of PT in reducing UI and increasing quality of life (QOL) in people with MS.
The term UI includes both stress and urge incontinence. Stress incontinence occurs when the pelvic floor muscles (PFMs) are too weak to stop urine from leaking when coughing, laughing, or sneezing.13 Urge incontinence or overactive bladder (OAB) occurs when urine leakage is closely preceded by a powerful urge to pass urine.13 In the United Kingdom, OAB and lower urinary tract symptoms (LUTS) are predominant in the aging population.13 The LUTS include frequency, urgency, stress, and urge UI as well as voiding or obstructive complications. The annual health-care and indirect costs in 2004 in the United States for UI were $19.5 billion and for OAB were $12.6 billion,5,14 which is projected to increase to $76.2 billion in 2015.7The most prevalent types of incontinence in MS are LUTS and OAB, affecting approximately 32% to 96% and 60% to 80%, respectively.15–18 This may be underreported if, as with the general population, people with MS wait an average of 6.5 years after the onset of symptoms before seeking treatment.6,8
The embarrassing nature of UI affects people's QOL in multiple ways, as measured using the 36-item Short Form Health Status Survey (a general health survey).10,19 The greatest deficits are seen in the subcategories of physical and social functioning, emotional health, and role limitations.10 Also, OAB was associated with depression and low self-esteem along with reduced activities of daily living (ADLs).10 With reduced activity, deconditioning may occur.2,8,10 Deconditioning has detrimental effects not only on physical structures (ie, cardiovascular and digestive systems) but also on the likelihood of depression and subjective low QOL.15,18,20 Although the impact of UI on QOL has been reported mostly in the general population, the effect in MS is likely similar.
The management of incontinence and its symptoms may be conservative or surgical. Although not all of these methods are routinely used clinically, conservative approaches described in the literature include clean intermittent self-catheterization or permanent indwelling catheters, anticholinergic agents, desmopressin, cannabinoids, complementary and alternative medicine, dorsal penile/clitoral nerve stimulation, and percutaneous posterior tibial nerve stimulation.9,21,22 Minimally invasive surgical treatments involve sacral neuromodulation,22,23 botulinum toxin injections, and augmentation cystoplasty.9 The most commonly used management methods are anticholinergic therapies, followed by clean intermittent self-catheterization or permanent indwelling catheters. Catheters are usually reserved for patients with bladder atonia or underactive bladder. The risk of infection with the use of catheters is compounded by the risk of hematuria and bladder or renal stones, as well as the hazard of urethral erosions in the long term.9
Anticholinergic agents have limited success, lack efficacy, and are not well tolerated in the long term by people with MS. 9,24,25 Their documented adverse effects include constipation, xerophthalmia, and xerostomia. Incomplete voiding is also increased with the use of these drugs,9,24 exacerbating existing symptoms and resulting in low compliance.
A primary conservative strategy to aid with incontinence is PFM training (PFMT), developed by Dr. Arnold Kegel in 1948 for use in stress UI.13 The rationale is to increase voluntary contraction of the PFMs before events such as coughing to reduce urinary leakage.11,12 Strengthening of the PFMs is also thought to help prevent adverse perineal movement during intra-abdominal pressure changes.26,27 A possible disadvantage of overtraining the PFMs is an increase in voiding disorders.28 This can increase bladder dysfunction and decrease control of PFM relaxation, which results in problems such as hesitancy, intermittence of stream, and high postvoid residuals.27,28 Residuals of urine left in the bladder increase the risk of infection.
Neuromuscular electrical stimulation (NMES) and electromyography (EMG) biofeedback are PT interventions that teach the patient to control voluntary muscle relaxation and contraction through visual and auditory feedback. Motor learning of these voluntary muscle techniques is achieved by high session frequency.28 Burgio et al.29 studied 197 women with UI (but not MS) randomized into groups: biofeedback-assisted behavioral treatment, drug treatment, or a placebo control condition. The biofeedback group showed an 80.7% reduction in incontinence episodes, significantly different from drug treatment (68.5% reduction). Both were more effective than the placebo control condition (39.4% reduction).29 Actual evidence of changes in UI for people with MS is needed before advocating PT interventions widely.11,12
The cost of this condition, financially and in QOL,3,25 highlights the need for a safe, well-tolerated, and effective long-term treatment that may preclude the need for surgical intervention in people with MS. Tubaro et al.,9 in a review of the management of incontinence in MS, made no discernible or definitive conclusions as treatment guidelines; standardization was hampered because of the heterogeneity of MS progression and bladder dysfunction.9,25 To our knowledge, no meta-analysis has specifically reviewed PT interventions and increased QOL related to incontinence in the MS population.
Question and Hypothesis
This review asks whether PT interventions decrease UI and improve QOL in people with MS. The null hypothesis is that PT interventions do not improve QOL or decrease UI in people with MS. It is alternatively hypothesized that PT interventions provide a statistically significant improvement in QOL and decrease UI in people with MS. The premise is that PT interventions are safer and better tolerated than drugs taken over the long term because there are no adverse effects of drug interactions with MS prescriptions,3 and treatments can be more readily adapted and tailored to each individual.
Methods
Inclusion and Exclusion Criteria
The inclusion of articles in this review was based on the following criteria: 1) participants had a diagnosis of MS; 2) participants had UI/OAB/urgency; 3) the intervention was performed by a PT, focusing on UI or bladder dysfunction; 4) the outcome measures included QOL and UI, assessed before and after intervention (for the within-group analysis) or compared with control groups (for the between-group analysis); and 6) the study was a level of evidence of 2b or better as defined by Jewell,30 or 4 of 10 on the Physiotherapy Evidence Database (PEDro) scale31 (a measure of the quality of reports of clinical trials). Articles were excluded if the interventions 1) included surgical or pharmaceutical treatment interventions, 2) focused on bowel incontinence, or 3) were not within the PT scope of practice.
Search Methods
A literature search was performed in the PubMed, Cumulative Index to Nursing and Allied Health Literature, PEDro, and Cochrane databases as well as in Google Scholar. The following search terms were used in combination and independently: incontinence, bladder dysfunction, urinary incontinence, multiple sclerosis, MS, physical therapy, physiotherapy, therapy, and rehabilitation. One researcher (V.B.) reviewed articles from the aforementioned databases for studies meeting the criteria. A recursive search of the references from relevant articles was completed, as well as a broad Google Scholar search to ensure that no relevant articles were missed. Two secondary reviewers (M.R., M.M.) verified that the selected articles met the criteria. If an article was not found on the PEDro, the PEDro scale score was calculated by the primary author (V.B.) (using the original PEDro scale score sheet)31 and was verified by a secondary reviewer (D.D.A.).
Statistical Analysis
Data regarding changes in QOL and UI were extracted from articles retrieved. The QOL and UI measures were categorized as follows: 1) participation QOL included questionnaires focused on the participation level in the patients' social environment, such as “How has urine leakage affected your participation in social activities outside of your home?” (Incontinence Impact Questionnaire-7 [IIQ-7], where 0 = not at all and 3 = greatly)32; 2) activity QOL included questionnaires focused on the effect of UI on task accomplishment, such as the amount of bother the person feels doing ADLs as a result of “frequent urination” (Urinary Distress Inventory-6 [UDI-6]: 0 = not at all bothered and 4 = greatly bothered)32; 3) objective measures of functional control mechanisms, such as EMG; and 4) recorded episodes of incontinence or leakage episodes, which includes 24-hour pad tests and bladder diaries. The reliability of the IIQ-7 and the UDI-6 have not been reported, although they have been used in multiple studies to record the impact of bladder dysfunction.9
Individual effect sizes for each category of outcome measure were combined to calculate the grand statistics, and a 95% confidence interval was calculated. The data are presented in forest plots of within- and between-study statistics. The Q heterogeneity statistic was calculated for each meta-analysis; where Q was large (P < .05), the random effects model was used to calculate the combined effect sizes. To convert the within-group effect size to clinical units, the standard deviation before the intervention was multiplied by the grand effect size. For the between-group conversion into clinical units, the between-group pooled standard deviation was multiplied by the grand effect size.
Results
Description of the Studies
Three hundred ninety-eight records were identified through a search of electronic article databases through November 26, 2013 (Figure 1). After removing duplicates and irrelevant records, 42 full-text articles were reviewed for eligibility. Of these 42 articles, 7 met the inclusion criteria. One of these studies was a systematic review and was used as a background reference because it reported no statistical data. Six nonoverlapping articles were used for data analysis. Their PEDro scale scores were all 4 of 10 or greater (Table 1).31 The articles used in this review are outlined in the following paragraphs and are summarized in Table 2.
Figure 1. PRISMA (Preferred Reporting Items of Systematic Reviews and Meta-analyses) diagram outlining article selection
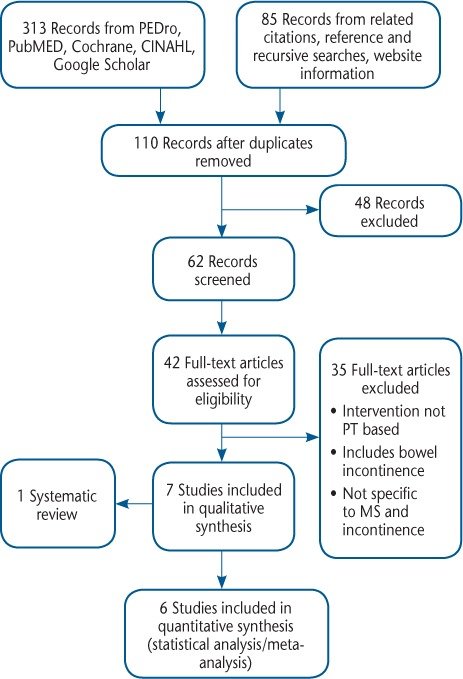
Table 1 Level of evidence of studies used in statistical analysis
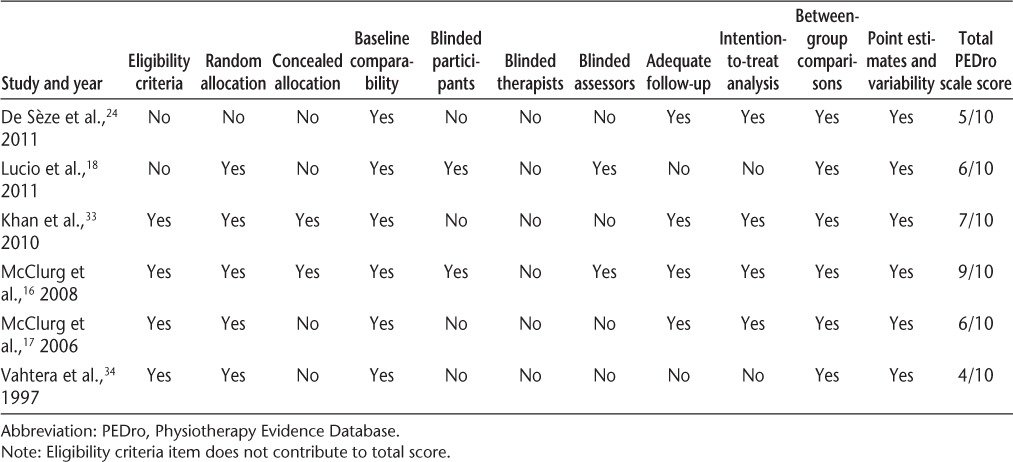
Table 2 Summary of studies
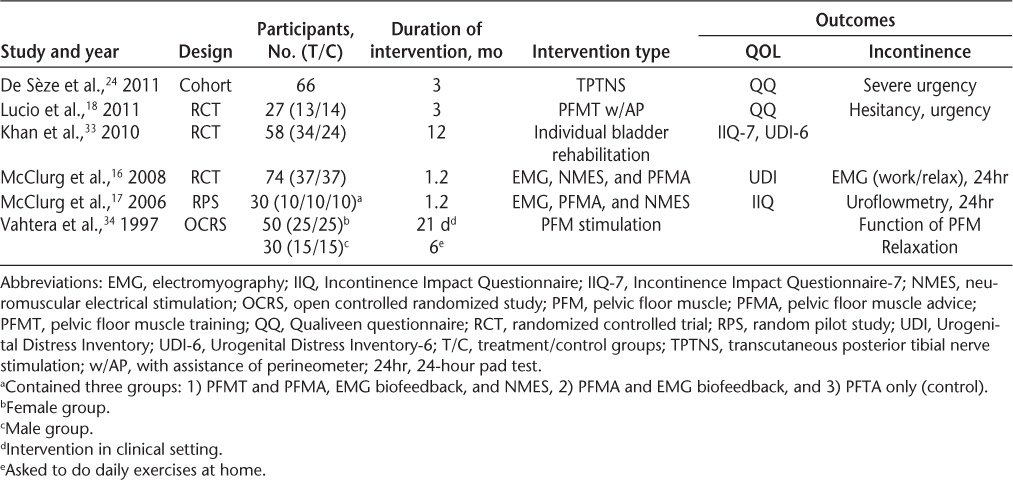
Lucio et al.18 included 27 females with MS and LUTS in a randomized controlled trial to determine the effects of PFMT on incontinence and QOL. Participants were blindly assigned to either a control group or a treatment group, and pre-post measures of incontinence (storage, voiding, etc.) and QOL scores were recorded by blinded PTs. Treatment groups underwent PFMT with assistance from a vaginal perineometer twice a week and were instructed to perform daily exercises at home. Control group treatment consisted of only introduction of the perineometer into the vagina for 30 minutes. Outcome measures consisted of five QOL measures and subjective concerns about storage and voiding symptoms. The authors reported no differences in baseline demographic data or in compliance with session attendance between groups. The researchers found improvement in LUTS and a positive effect on QOL in women who underwent PFMT treatment.18
Khan et al.33 conducted a randomized controlled trial studying the outcomes of bladder rehabilitation in 58 women with MS. They randomized participants into either a treatment group (n = 24) or a waitlist control group (n = 34). The treatment group received an inclusive, personalized, multidisciplinary rehabilitation program (PT, occupational therapy, speech pathology, and social work) in either an inpatient or an outpatient setting two to three times per week for 6 weeks. Thereafter, they continued with a maintenance program (stretching and home exercises) similar to the ones undertaken by the control group. Twelve months after the intervention ended, the treatment group was reassessed: QOL participation (IIQ-7) and activity (UDI-6) outcome measures showed statistically significant improvements compared with the control group. The control group was reassessed 12 months after the baseline assessment. Khan et al.33 did not allocate specific interventions to be used for the various types of bladder dysfunction seen in MS (OAB, urinary retention, detrusor sphincter dyssynergia, etc.).
Vahtera et al.34 studied the effectiveness of pelvic floor rehabilitation in people with MS. Fifty females and 30 males were divided randomly into either a control or a treatment group, with similar baseline demographic features. Treatment consisted of six electrical stimulation sessions in 21 days, and pelvic floor exercises were prescribed to do at home daily for at least 6 months. The control group received no treatment. All the participants were assessed at 3 weeks, 2 months, and 6 months. Significantly positive results were presented for the symptoms of urinary urgency, frequency, and incontinence, although no difference was noted during voluntary PFM relaxation between baseline and discharge for EMG activities. Notably, male participants responded better to the treatment than females. The authors concluded that electrical stimulation together with PFM exercises constitutes an effective treatment for LUTS in males with MS. They made no definitive conclusions regarding the effectiveness of this treatment in females.
In 2006, McClurg et al.17 conducted a randomized pilot study to compare PFMT and PFM advice (PFTA), EMG biofeedback, and NMES for bladder dysfunction in people with MS. Thirty females were randomly assigned to one of three groups: 1) PFTA, 2) PFTA and EMG biofeedback, or 3) PFTA, EMG biofeedback, and NMES. All the participants underwent 9 weeks of therapy. Outcome measures included 24-hour pad test, digital assessment of the pelvic floor (to evaluate power, endurance, number of repetitions, and number of fast contractions of the PFM, scaled from 0 to 5, with 0 being the lowest and 5 being the highest score), EMG biofeedback contraction and relaxation, voiding data, and QOL questionnaires (IIQ and UDI).17 The authors concluded that the combination of PFTA, EMG biofeedback, and NMES can reduce urinary symptoms in people with MS. In a separate study in 2008, McClurg et al.16 studied the effects of NMES on the treatment of LUTS in MS using a double-blind, placebo-controlled, randomized clinical trial. Of the 74 patients enrolled, the control group, with 26 women and 11 men, received PFMT, EMG biofeedback, and a placebo NMES. The treatment group, with 31 women and 6 men, received active NMES, PFMT, and EMG biofeedback. The results were not reported separately for men and women. Both groups were given general strategic advice regarding the maintenance of symptoms and were instructed to perform daily PFM exercises. Outcome measures were collected at baseline and at weeks 9 (end of intervention), 16, and 24. Statistical analysis between groups showed significant differences at each time point in the following test results: the 24-hour pad test, the digital assessment of PFMs (Oxford classification), and the IIQ and UDI. The EMG data regarding contraction, relaxation, and endurance showed no significant changes. McClurg et al.16,17concluded that a combination of PFMT, NMES, and EMG biofeedback should be used as a “first-line option in alleviating some symptoms of LUTS associated with MS.”16(p231)
De Sèze et al.24 studied transcutaneous posterior tibial nerve stimulation (TPTNS) for the treatment of OAB in MS using results from a multicenter prospective study; TPTNS is a noninvasive technique of peripheral electrical neuromodulation using surface electrodes. It modulates the sacral nerve plexus indirectly via the posterior tibial nerve, a branch of the sciatic nerve, which originates from the same segment in the spine as the nerves to the bladder and pelvic floor.35 The exact mechanisms of action are not known; however, positive results have been observed in treating OAB and UI as well as fecal incontinence.35 Seventy patients, 51 women and 19 men (final n = 66: 4 were lost to follow-up; sex was not specified) were evaluated in a single-group design, and TPTNS was applied unilaterally via electrodes behind the right medial malleolus. Patients were taught to use the device at home for 20 minutes daily for 3 months. Results of primary and secondary outcome measures were recorded at baseline, 30 days, and 90 days. Use of TPTNS statistically significantly reduced urgency and had a positive effect on QOL scores (Qualiveen questionnaire). It is unknown whether these effects were maintained long term.
Data Analysis
The combined effect sizes and 95% confidence intervals were calculated for individual studies and across studies (Table 3).36 These data are represented in a forest plot in Figure 2.37 Where Q was large (P < .05), the random effects model38 was used (Table 3).
Figure 2. Forest plots
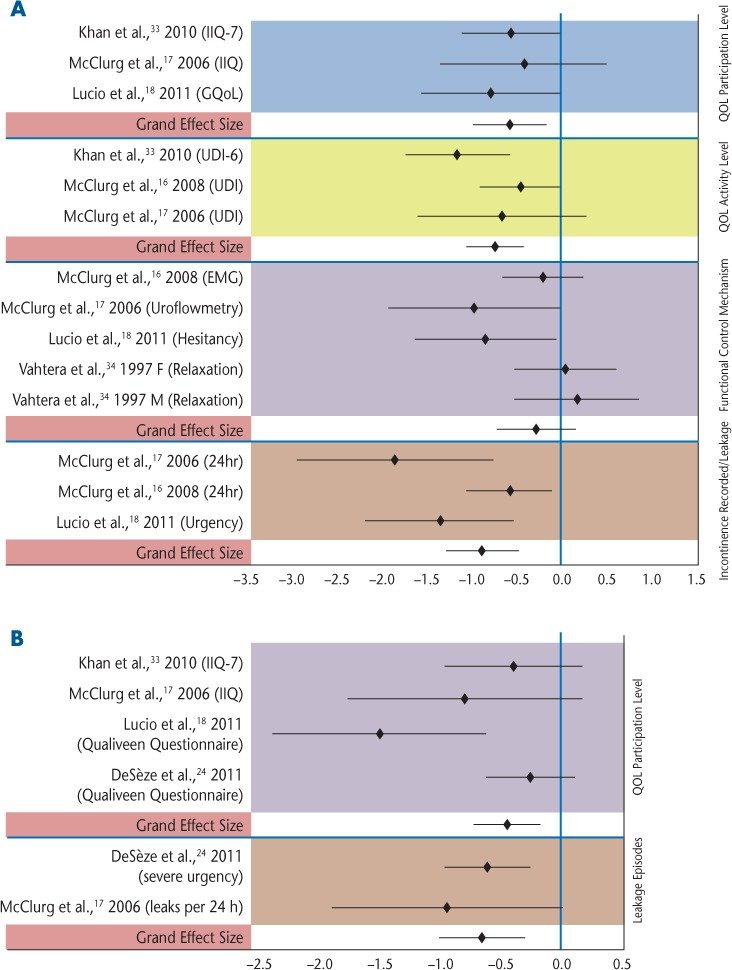
Table 3 Summary of within- and between-group combined effect sizes and 95% CIs
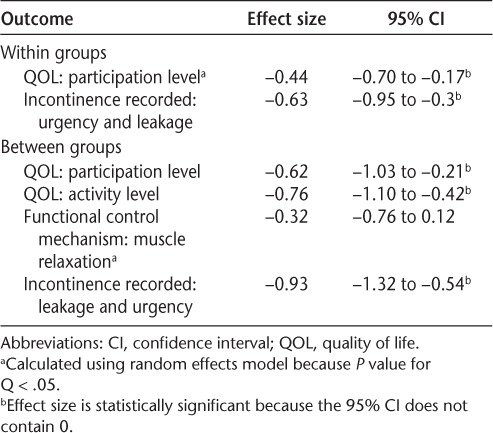
A within-group analysis of QOL participation level was performed for the studies by Khan et al.,33 McClurg et al.,17 De Sèze et al.,24 and Lucio et al.18 Analysis of incontinence recorded (urgency and leaks per 24 hours) was performed using data from De Sèze et al.24 and McClurg et al.17 Figure 2B portrays these analyses, showing the grand effect sizes to be statistically significant for both outcome measures.
Between-group analyses for all the outcome measures are depicted in Figure 2A. Grand effect sizes were statistically significant for QOL and incontinence leakage episodes but not for functional control mechanisms (eg, EMG data on strength, relaxation, and endurance).
Harm
De Sèze et al.24 reported no adverse effects relating to the use of TPTNS, confirming findings from previous studies as to the safety of this treatment. The other studies in the present review affirmed high adherence rates and good tolerance for the treatments used.16,17,33,34 No adverse effects were reported.
Cost
There were no data available for the individual cost of each PT session for the various treatments. McClurg et al.16 suggested that with a 50% reduction in the use of medication or containment, there was a possibility of a £220.00 direct savings per individual over 6 months. All the authors reported an increase in QOL for people with MS; however, this was not quantified in financial terms.
Discussion
The aim of this review was to determine whether the current literature supports the effectiveness of PT as a treatment for UI and QOL in people with MS. Six studies met the inclusion criteria and had sufficient statistical data for a meta-analysis. Within-study analysis showed statistical significance for the incontinence leakage episodes and QOL participation level. The between-study analysis displayed significance for QOL outcomes and urinary leakage episodes. Functional control mechanisms did not reach statistical significance, although a positive difference was observed between groups. Overall, these results allow us to reject the null hypothesis that PT interventions do not improve QOL or UI in people with MS, leaving the alternative hypothesis that PT interventions present a statistically significant improvement in at least some measures of QOL and UI.
To assess the clinical relevance of the between-group results, effect sizes were converted to clinical units using the pooled standard deviation between the experimental and control groups. In terms of QOL participation level according to McClurg et al.,17 the average effect size corresponds to an IIQ score difference between groups of 32 points. On this scale, a score of 0 represents “least bother,” and 100 represents the “greatest bother.”32 Although no minimal clinically important difference has been published for this measure, maintenance of QOL in populations with a degenerative disease is advantageous. In terms of QOL activity level, the average effect size was statistically significant, and it may be clinically important, with an average difference of 43 points in the mean UDI score17 between groups. This number surpasses the anchor-based minimal important difference of −22.4 that Barber et al.39 calculated in a general population. Clinical conversion of the effect sizes for incontinence leakage episodes was not possible owing to lack of reported data.
For the within-group analysis, in terms of leakage episodes, according to McClurg et al.,17 on average patients experienced 2.1 fewer leaks per 24 hours (average of 50% decrease) after treatment with EMG, PFM advice, and NMES. In terms of participation QOL, according to McClurg et al.,17 there was an average decrease from before to after the intervention of 36 points on the IIQ. There are no minimally clinically important differences known for these outcomes in this population.
Physical therapy interventions in the included studies resulted in a direct effect on incontinence leakage episodes, as seen in the significant grand effect sizes within (−0.63) and between (−0.93) groups. The moderate effect sizes in QOL measures may indirectly result from fewer leakage episodes, but multiple factors can contribute to changes in QOL. For example, fatigue, muscle weakness, time required for ADLs, and depression can result in a decrease in QOL participation and activity levels.1 Change in any of these factors may contribute to improvements seen in this sample of participants. In addition, the decrease in UI can, in turn, reduce fatigue and depression, amplifying the effect on QOL.
The studies reported heterogeneity in the qualitative aspect of their designs. Differences in the numbers of participants and the types of interventions may have affected the results. Objective measures of functional control mechanisms (EMG/relaxation) did not reach statistical significance, possibly owing to differences in treatment frequencies (5 weeks in both studies by McClurg et al.,16,17 and only 3 weeks for the intervention in the study by Vahtera et al.34). The longer-duration treatments were associated with greater effect sizes. Other factors that may have contributed to heterogeneity include different placement of the EMG electrodes and lower adherence in the study by Vahtera et al.34 McClurg et al.16 cited high adherence values for their population.
Limitations
There were several limitations in this study that may have affected the results. 1) Although care was taken to use relevant search terms, and a variety of search engines, some literature may have been missed. 2) The sample sizes for individual studies were fairly small, which decreases the power of the individual studies, but the grand effect sizes for most of the outcomes were significant when the studies were combined. 3) Results may be limited due to the heterogeneity of MS type as well as type of incontinence in the sample populations. MS is a degenerative and unpredictable disease, hindering formulation of a standard treatment plan for everyone. A benefit of PT interventions may be that the dose, duration, and combination of treatments can be easily modified for each individual. For example, McClurg et al.17 described the amount of total EMG biofeedback treatment duration as depending on fatigue. 4) Most of the studies lacked a retention follow-up test to assess the long-term change. McClurg et al.16 observed maintenance of reduced leakage episodes, excellent adherence, and motivation until the end of their observation at 24 weeks. The authors reported that motivation and adherence have been shown to have a strong association with outcome in previous studies.16,29
This meta-analysis included all types of PT interventions, restricting our ability to identify specific interventions that deliver the greatest effects. Physical therapy interventions do decrease incontinence and increase QOL. They are well tolerated and safe for people with MS, as no adverse or harmful effects were reported during the studies.
Future Directions
Future studies should look at specific types of MS with different forms of incontinence to determine the types of treatment that are most effective and efficient for each demographic feature. Examination of specific PT interventions with larger sample sizes could provide clinical practice guidelines for treating people with MS through conservative means. Longer follow-up studies would help determine the lasting effects of PT interventions and would shed light on the learning aspects of the treatment protocols. Data published by the National Association for Continence6 show that patients with incontinence are largely unaware of the options and benefits of PT interventions. Published data from future studies could help solidify the benefits of nonpharmaceutical or surgical interventions for this population.
Conclusion
Incontinence is a prevalent problem in the MS population and in the general public. Physical therapy interventions can help reduce the negative effects of UI by decreasing some of the symptoms and increasing QOL. Treatment protocols vary, lacking a definitive technique. With further research, specific intervention methods and outcomes could be improved.
Footnotes
From the Department of Physical Therapy and Rehabilitation Science, University of California San Francisco/San Francisco State University, San Francisco, CA, USA.
Financial Disclosures: The authors have no conflicts of interest to disclose
References
1. Nicholas R, Rashid W. Multiple sclerosis. Am Fam Physician. 2013;87:712–714. [PubMed]
2. Coote S, Hogan N, Franklin S. Falls in people with multiple sclerosis who use a walking aid: prevalence, factors, and effect of strength and balance interventions. Arch Phys Med Rehabil. 2013;94:616–621.[PubMed]
3. National Multiple Sclerosis Society website. http://www.nationalms-society.org. Accessed November 20, 2013.
4. Holland NJ. Urinary Dysfunction and Multiple Sclerosis: A Clinical Practice Guideline for Professionals Developed by the Multiple Sclerosis Council for Clinical Practice Guidelines. New York, NY: National Multiple Sclerosis Society; 2013.
5. Markland AD, Richter HE, Fwu CW, Eggers P, Kusek JW. Prevalence and trends of urinary incontinence in adults in the United States, 2001 to 2008. J Urol. 2011;186:589–593. [PMC free article] [PubMed]
6. National Association for Continence. National Association for Continence facts and statistics.http://www.nafc.org/index.php?page=facts-statistics. Accessed November 7, 2013.
7. Milsom I, Coyne KS, Nicholson S, Kvasz M, Chen C-I, Wein AJ. Global prevalence and economic burden of urgency urinary incontinence: a systematic review. Eur Urol. 2014;65:79–95. [PubMed]
8. Khan F, Turner-Stokes L, Ng L, Kilpatrick T. Multidisciplinary rehabilitation for adults with multiple sclerosis. Cochrane Database Syst Rev. 2007;(2):CD006036. [PubMed]
9. Tubaro A, Puccini F, De Nunzio C. et al. The treatment of lower urinary tract symptoms in patients with multiple sclerosis: a systematic review. Curr Urol Rep. 2012;13:335–342. [PubMed]
10. Tubaro A. Defining overactive bladder: epidemiology and burden of disease. Urology. 2004;64(suppl 1):2–6. [PubMed]
11. Temml C, Haidinger G, Schmidbauer J, Schatzl G, Madersbacher S. Urinary incontinence in both sexes: prevalence rates and impact on quality of life and sexual life. Neurourol Urodyn. 2000;19:259–271.[PubMed]
12. Paltamaa J, Sjogren T, Peurala SH, Heinonen A. Effects of physiotherapy interventions on balance in multiple sclerosis: a systematic review and meta-analysis of randomized controlled trials. J Rehabil Med.2012;44:811–823. [PubMed]
13. National Institute for Health and Care Excellence. Urinary incontinence: introduction: a general introduction to urinary incontinence. http://www.evidence.nhs.uk/topic/urinary-incontinence. Accessed November 15, 2013.
14. Hu T, Wagner TH, Bentkover DJ, Leblanc K, Zhou SZ, Hunt T. Costs of urinary incontinence and overactive bladder in the United States: a comparative study. Urology. 2004;63:461–465. [PubMed]
15. Seth JH, Sahai A, Panicker JN. Lower urinary tract dysfunction in multiple sclerosis. Curr Bladder Dysfunct Rep. 2012;7:97–104.
16. McClurg D, Ashe RG, Lowe-Strong AS. Neuromuscular electrical stimulation and the treatment of lower urinary tract dysfunction in multiple sclerosis: a double blind, placebo controlled, randomised clinical trial. Neurourol Urodyn. 2008;27:231–237. [PubMed]
17. McClurg D, Ashe RG, Marshall K, Lowe-Strong AS. Comparison of pelvic floor muscle training, electromyography biofeedback, and neuromuscular electrical stimulation for bladder dysfunction in people with multiple sclerosis: a randomized pilot study. Neurourol Urodyn. 2006;25:337–348. [PubMed]
18. Lucio AC, Perissinoto MC, Natalin RA, Prudente A, Damasceno BP, D'ancona CA. A comparative study of pelvic floor muscle training in women with multiple sclerosis: its impact on lower urinary tract symptoms and quality of life. Clin Sci. 2011;66:1563–1568. [PMC free article] [PubMed]
19. Milsom I, Abrams P, Cardozo L. et al. How widespread are the symptoms of an overactive bladder and how are they managed? a population-based prevalence study. BJU Int. 2001;87:760–766. [PubMed]
20. Robinson D, Cardozo L. Overactive bladder: diagnosis and management. Maturitas. 2012;71:188–193.[PubMed]
21. Sevilla GS. Are cannabinoids more effective than placebo in decreasing MS-related bladder dysfunction? Br J Neurosci Nurs. 2012;8:71–78.
22. Minardi D, Muzzonigro G. Lower urinary tract and bowel disorders and multiple sclerosis: role of sacral neuromodulation: a preliminary report. Neuromodulation. 2005;8:176–181. [PubMed]
23. Ruud Bosch JL, Groen J. Treatment of refractory urge urinary incontinence with sacral spinal nerve stimulation in multiple sclerosis patients. Lancet. 1996;348:717–719. [PubMed]
24. De Sèze M, Raibaut P, Gallien P. et al. Transcutaneous posterior tibial nerve stimulation for treatment of the overactive bladder syndrome in multiple sclerosis: results of a multicenter prospective study. Neurourol Urodyn. 2011;30:306–311. [PubMed]
25. Abernethy DA, Weatherall M, Siegert RJ, Hay-Smith EJC. Bladder and bowel dysfunction in multiple sclerosis: a review of treatment effectiveness. Aust N Z Continence J. 2007;13:81–82. 85, 87–89.
26. Dannecker C, Wolf V, Raab R, Hepp H, Anthuber C. EMG-biofeedback assisted pelvic floor muscle training is an effective therapy of stress urinary or mixed incontinence: a 7-year experience with 390 patients. Arch Gynecol Obstet. 2005;273:93–97. [PubMed]
27. Dumoulin C, Glazener C, Jenkinson D. Determining the optimal pelvic floor muscle training regimen for women with stress urinary incontinence. Neurourol Urodyn. 2011;30:746–753. [PubMed]
28. Doggweiler-Wiygul R, Sellhorn E. Role of behavioral changes and biofeedback in urology. World J Urol. 2002;20:302–305. [PubMed]
29. Burgio K, Locher J, Goode P. et al. Behavioral vs drug treatment for urge urinary incontinence in older women: a randomized controlled trial. JAMA. 1998;280:1995–2000. [PubMed]
30. Jewell DV. Guide to Evidence-Based Physical Therapy Practice. Sudbury, MA: Jones and Bartlett; 2008.
31. PEDro scale. PEDro Physiotherapy Evidence Database website.http://www.pedro.org.au/english/downloads/pedro-scale. Accessed November 30, 2013.
32. Harvey MA, Kristjansson B, Griffith D, Versi E. The Incontinence Impact Questionnaire and the Urogenital Distress Inventory: a revisit of their validity in women without a urodynamic diagnosis. Am J Obstet Gynecol. 2001;185:25–31. [PubMed]
33. Khan F, Pallant JF, Pallant JI, Brand C, Kilpatrick TJ. A randomised controlled trial: outcomes of bladder rehabilitation in persons with multiple sclerosis. J Neurol Neurosurg Psychiatry. 2010;81:1033–1038.[PubMed]
34. Vahtera T, Haaranen M, Viramo-Koskela AL, Ruutiainen J. Pelvic floor rehabilitation is effective in patients with multiple sclerosis. Clin Rehabil. 1997;11:211–219. [PubMed]
35. Booth J. Institute for Applied Health Research, Glasgow Caledonian University; A pilot trial of transcutaneous posterior tibial nerve stimulation for bladder and bowel dysfunction in older adults in residential care. http://www.rcn.org.uk/__data/assets/pdf_file/0017/512234/2013_RCN_research_6.3.2.pdf. Accessed January 21, 2014.
36. Hedges L, Shymansky J, Woodworth G. A Practical Guide to Modern Methods of Meta-analysis.Washington, DC: National Science Teachers Association; 1989.
37. Clark O, Djulbegovic B. Forest plots in Excel software (data sheet) http://www.evidencias.com.
38. Borenstein M, Hedges L, Rothstein H. Meta-analysis: fixed effect vs. random effects model. Introduction to Meta-analysis.http://www.metaanalysis.com/downloads/Meta%20Analysis%20Fixed%20vs%20Random%20effects.pdf.
39. Barber MD, Spino C, Wheeler TL. The minimum important differences for the urinary scales of the Pelvic Floor Distress Inventory and Pelvic Floor Impact Questionnaire. Am J Obstet Gynecol. 2009;200:e1–e7. [PMC free article] [PubMed]
Articles from International Journal of MS Care are provided here courtesy of The Consortium of Multiple Sclerosis Centers
