Multiple Sclerosis in Pregnancy
Why is this important to me?
MS is frequently diagnosed in women in their childbearing years. Thus, you may find that the desire to have a baby and the need to manage your MS overlap. Pre-pregnancy family planning, the risks and benefits of taking drugs for MS before, during, and after pregnancy, and other pregnancy-related issues should be discussed with your healthcare provider.
Who will benefit from reading this study/article?
People with MS who are pregnant or considering becoming pregnant will benefit from reading this article.
What is the objective of this study?
This article reviewed several issues that people with MS are likely to encounter before, during, and after pregnancy.
Things to consider before you become pregnant:
- Pregnancy does not affect long-term MS progression.
- The various hormones and other physiological changes that occur during pregnancy may make you less likely to experience a relapse while pregnant.
- Mitoxantrone (Novantrone), which is one drug used to treat MS, can lead to menopause.
- Men with MS may have lower sperm quality and quantity.
- A child who has a parent with MS has a 3-4% risk of developing the disease compared to a 0.1-0.2% risk in the general population.
- Whether and when to stop taking MS medication before and during pregnancy is a decision for each individual patient and involves input from you, your neurologist, and your obstetrician.
Things to consider while you are pregnant:
- If you have MS, preterm birth rates are not higher than in the general population.
- If you have a tendency to develop urinary tract infections, these infections may increase during pregnancy, and if you use a wheelchair or have paralysis, this risk may further increase during pregnancy.
- Pregnancy increases your risk of falling if you have MS. Maintaining a healthy pregnancy weight gain, maintaining your range of motion, performing stretching exercises, and using a walker or wheelchair may decrease the risk of falling.
Things to consider regarding delivery of your baby:
- That said, patients with MS may have a greater tendency to require labor induction, a longer second stage of labor, a higher risk of the need for an operative vaginal birth, a higher risk of cesarean delivery, and a lower infant birth weight. The decision to pursue vaginal or cesarean delivery is based on your obstetric needs and not your MS.
- Study results are conflicting regarding whether pain medication during delivery impacts the incidence of MS relapses after delivery.
Things to consider after your baby is born:
- Immediately after your baby is born, you may experience extreme fatigue, worsening MS symptoms, and an increased tendency to fall. If needed, disease-modifying therapies and steroids can be started immediately after your baby is born to control your symptoms
- You have an increased risk of relapse during the first 3-6 months after delivery.
- Evidence suggests that breastfeeding may reduce the risk of relapse by about 50%. However, the evidence does not provide a clear recommendation on whether you should take your MS medication while breastfeeding. For this reason, it is important that you discuss this decision with your neurologic and obstetric health care providers.
- If you do not plan to have another baby soon, hormonal contraception (e.g., the pill) is generally considered safe to use if you have MS and does not negatively impact MS prognosis or disability severity. The current recommendations from The American Congress of Obstetricians and Gynecologists (ACOG) include the IUD, as well as the pill.
How did the authors study this issue?
The authors reviewed current recommendations about issues related to pregnancy in women with MS.
| SHARE: | |||||
Original Article
Multiple Sclerosis in Pregnancy
The Journal of Perinatal & Neonatal Nursing
Suzanne McMurtry Baird, DNP, RN; Jennifer Dalton, MSN, RN
ABSTRACT
Multiple sclerosis is the most common chronic neurologic disability in young adults in their childbearing ages of 20 to 45. The disease affects more women than men, which prompts discussion of pregnancy-related issues in a woman with multiple sclerosis. Preconceptual counseling to discuss the safety of medications in pregnancy, the antepartum period along with what the patient can expect during birth, and the postpartum period will be discussed.
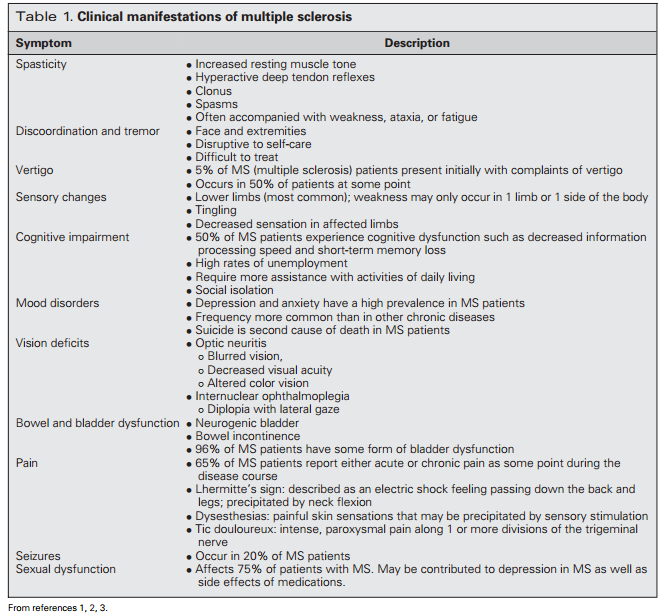
Multiple sclerosis (MS) is a progressive degenerative disease involving the myelin sheath of neurons in the central nervous system. Multiple sclerosis is caused by an autoimmune response involving macrophages and cytotoxic T cells that cause a chronic inflammatory state resulting in damage to the myelin sheath around nerve cells. The demyelination of axons causes both acute and chronic lesions to develop in the central nervous system altering the conduction of electrical impulses to muscle groups. Multiple sclerosis is a progressive disease with varying symptoms based on the location of lesions. Vision, speech, muscle strength, and sensation are commonly affected by MS. Physical symptoms can be accompanied by memory loss, cognitive impairment, depression, mood swings, and fatigue.1−3 Table 1 describes these clinical manifestations.
Exacerbation of MS symptoms can be precipitated by infection, stress, pregnancy (postpartum period), fatigue, heat, and heavy metal exposure.3 The purpose of this article is to discuss preconception care for women with MS; potential pregnancy issues; education concerns before, during, and after pregnancy; and anticipatory management of intrapartum and postpartum needs.
PRECONCEPTION ISSUES AND COUNSELING
Multiple sclerosis is the most common chronic neurologic disability in young adults of childbearing age, with nearly twice as many women than men diagnosed.1,2,4 The specific cause of MS is unknown but is believed to be caused by a combination of several factors including immunologic, genetic, and environmental factors. Previously, pregnancy was discouraged in women with MS; however, recent studies have shown pregnancy as potentially having a beneficial role on MS relapse rates with no effects to long-term progression of the disease.5−8
Physiologic changes during pregnancy cause a down-regulation of cellular immune responses and an increase in production of neuroprotective hormones. Pregnant women have high levels of circulating lymphocytes and macrophages, decreased production of nitric oxide, and production of proinflammatory cytokines such as interleukin-10.9 While other mechanisms may be involved, it is proposed that the estrogen increase during pregnancy plays a protective role as well.10 These immune system changes appear to have a cumulative, beneficial effect on MS by decreasing the incidence of a first clinical demyelinating event. In addition, there is evidence that with each additional pregnancy the incidence continues to decrease.10 The frequency of relapse during pregnancy drops, with the third trimester being the lowest. However, for 3 to 6 months after delivery the relapse rate significantly increases.7
Fertility and genetic risk
Women with MS often have concerns related to genetic risks and fertility. The genetic link to MS is not clearly understood. However, children of mothers with MS have a 3% to 4% risk of developing the disease compared with a 0.1% to 0.2% risk in the general population.11 If both parents are affected by the disease, the risk for offspring increases to 30.5%.12 Also known is that risk increases at 2% to 4% with a first-degree relative diagnosed with the disease.11 Recently, Ponsonby and colleagues10 showed that increasing gravidity, parity, and offspring number decreased the risk of a first clinical presentation of demyelination further supporting the protective effects of pregnancy. Currently, there are no diagnostic tests that predict MS.
There are no reports that show a direct influence of MS on fertility in women and/or their ability to conceive a pregnancy. However, there is uncertain clinical significance on fertility from one study showing women with MS having higher levels of prolactin, follicle-stimulating hormone, luteinizing hormone, total and free testosterone, 5-α dihydrotestosterone, δ-4 androstenedione levels, and decreased levels of estrone sulfate.13 In addition, mitoxantrone (Novantrone), used as a disease modifying medication, may induce early menopause in some women.12 Men with MS may have impaired fertility due to decreased sperm count, mobility, and normal sperm development.14
 Sexual dysfunction is often reported in chronic diseases and is associated with unhappiness and decreased quality of life.15 In women with MS, high levels of sexual dysfunction are reported with symptoms of weakness, spasticity, depression, bladder and bowel dysfunction, sleep disorders, pain, and sensory abnormalities, which may play a role in fertility.12,14,16
Sexual dysfunction is often reported in chronic diseases and is associated with unhappiness and decreased quality of life.15 In women with MS, high levels of sexual dysfunction are reported with symptoms of weakness, spasticity, depression, bladder and bowel dysfunction, sleep disorders, pain, and sensory abnormalities, which may play a role in fertility.12,14,16
If infertility occurs, women who undergo assisted reproductive technology with in vitro fertilization may be at increased risk for relapse of MS symptoms. There are no definitive data, but one small prospective study of 32 women with relapsing-remitting MS who underwent in vitro fertilization showed a 7-fold increase in the risk of MS exacerbation and a 9-fold increase in the risk of disease progression with new brain lesions observed in the 3 months following assisted reproductive technology.17 In another study, evidence of increased relapse rates were also demonstrated.18 Rapidly changing hormone cycles and/or exogenous hormone administration (gonadotropin releasing hormone) and failed in vitro attempts have been linked to the increase in MS activity for these women.19
Preconception medication management
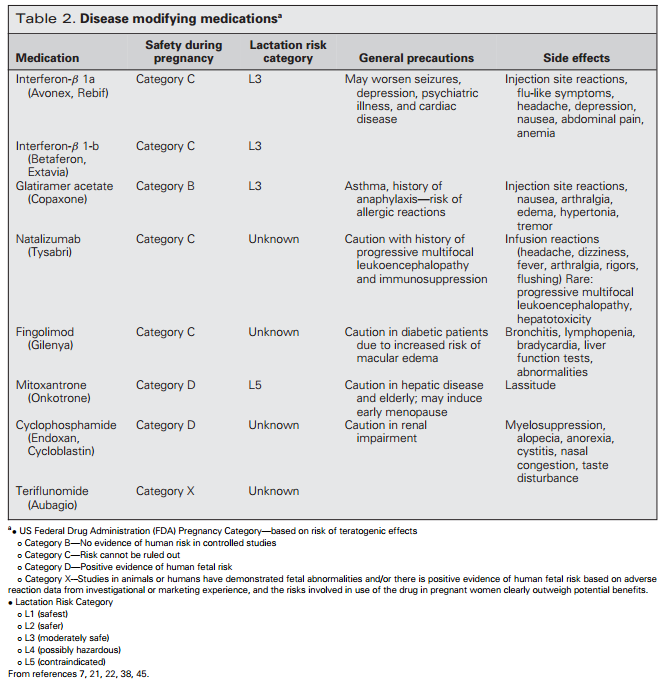
Many of the medications prescribed for MS are not recommended for use during pregnancy, requiring the woman and partner to plan the timing of pregnancy to adjust medications before conception and gain proper control of the disease. Table 2 outlines categories of disease-modifying medication and safety in pregnancy.
There are 8 disease-modifying medications approved by the Food and Drug Administration for the treatment of MS: glatiramer acetate, subcutaneous or intramuscular interferon β-1a, subcutaneous interferon β-1b, fingolimod, natalizumab, teriflunomide, and mitoxantrone (Novantrone). For the last decade, the first-line treatments for relapsing remitting MS have been interferon-β and glatiramer acetate.20 Both medications have shown a reduction in the number of active lesions by 30%.20 Secondline treatments include natalizumab or fingolimod.
Analysis of several large pregnancy registries suggest that first-line therapy agents, glatiramer acetate (FDA category B) and interferon (FDA category C), are not associated with teratogenic risk or a higher risk of miscarriage.21,22 These data were recently supported in a study evaluating 335 pregnant women with MS. Results showed congenital anomalies were within normal ranges in pregnancies exposed to interferon-beta and glatiramer acetate.21 However, the FDA has not changed the category rating for pregnancy risk on interferon-β.
Fingolimod is classified as a FDA category C medication based on animal studies and is the first oral treatment for relapsing-remitting MS. It is generally well tolerated and showed a 50% lower relapse rates than a comparison group taking placebo.20 Fingolimod is still being studied and the long-term safety has yet to be established. In addition to potential for teratogenic effects, fingolimod may take up to 2 months to be completely eliminated from the body.3 Therefore, contraception should be used during the 2 months following administration of this medication.
Natalizumab has shown reduction in the rate of disability progression, decreased relapse rate, and number of new lesions at a greater percentage than first-line treatments.20 Natalizumab is classified as a category C medication based on studies in guinea pigs. There are no well-controlled trials of natalizumab efficacy and safety in pregnant women. However, current registry data suggest that treatment has no apparent adverse effect on pregnancy outcomes in humans.3
Third-line treatments for patients with aggressive disease who show no improvement with other treatments include immunosuppression or chemotherapy, followed by autologous stem cell transplant.20 Mitoxantrone (Novantrone) is a cytotoxic chemotherapeutic agent used for secondary progressive MS. Use of mitoxantrone (Novantrone) during pregnancy has shown teratogenic effects in the developing fetus and intrauterine growth restriction at low doses in rats. Teriflunomide is a newer medication with a designated pregnancy category X. Teriflunomide has a long half-life (more than 2 weeks) and should only be prescribed to women using a reliable method of contraception.3
The decision of when to stop medication therapy is individualized and involves the woman, neurology, and obstetric providers, and it is usually based on disease activity in the months leading up to conception. On the basis of published evidence, women should be advised to stop their disease-modifying agents one menstrual cycle before attempting conception or if pregnancy is suspected.3 Additional data indicate no increase in teratogenic effects of natalizumab. After weighing the risks and benefits of the specific disease modifying agent and if required during pregnancy, mitoxantrone (Novantrone), fingolimod, and teriflunomide should be avoided.
Diagnostic criteria
Although onset of MS is uncommon during pregnancy, women’s healthcare providers need an understanding of diagnostic criteria to better discuss prognosis and pregnancy-related concerns. There are 4 clinical types of MS, varying from mild to severe. Relapsing-remitting MS is the most common, with 85% of people initially diagnosed with this type.3 It is characterized by exacerbations of worsening neurologic function that lasts at least 24 hours.3 Relapses usually begin with symptoms of neurologic deficits such as optic neuritis, asymmetric numbness, weakness, or ataxia that develops over several days.3 These symptoms are followed by remission in which no disease progression occurs. In 40% of attacks, mild residual symptoms remain during the remission.3
 Secondary-progressive MS occurs in about 40% to 45% of people diagnosed with relapsing-remitting MS after about 10 years.3 It is defined as a progressive accumulation of disability with no relapses.3 Temporary, minor improvements may occur but disease progression continues.
Secondary-progressive MS occurs in about 40% to 45% of people diagnosed with relapsing-remitting MS after about 10 years.3 It is defined as a progressive accumulation of disability with no relapses.3 Temporary, minor improvements may occur but disease progression continues.
In Primary-progressive MS, neurologic function slowly worsens over time without relapses or remissions. These patients may have an occasional plateau and temporary minor improvements in symptoms with return to a variable rate of progression.3 Ten percent of MS patients are diagnosed with this type.23
Progressive-relapsing MS is a rare form that occurs in only 5% of patients diagnosed. This subtype of MS is characterized by steadily worsening disease from onset with acute exacerbations and no remission periods.3
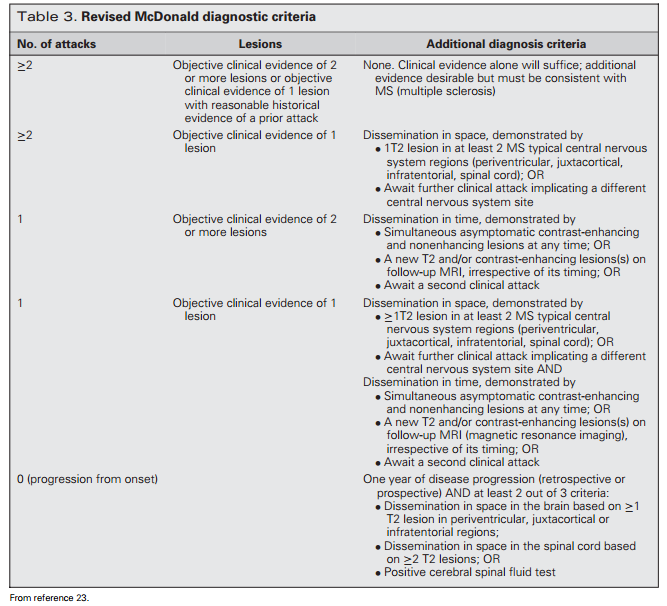
Progressive-relapsing MS usually presents with slow onset of spastic paraparesis followed by cerebellar or hemiplegic symptoms.20 Progressive-relapsing MS does not respond to disease modifying medications. The Revised McDonald Criteria is used to diagnose MS and are outlined in Table 3.23
The diagnosis includes evidence of lesions in at least 2 places in the central nervous system at least 1 month apart and differential diagnoses are ruled out. Differential diagnoses include migraine, cerebral neoplasms, nutritional deficiencies of vitamin B12 or copper, compressive lesions of the spinal cord, human immunodeficiency virus, syphilis, lupus, or psychiatric disease.20
ANTEPARTUM
Even with the positive hormonal influence that likely improves symptoms during pregnancy, women with prior diagnosis or who have continued symptoms/limitations due to MS are considered at increased risk. The design of prenatal care is individually determined and is based on the woman’s symptoms and/or physical disabilities. To monitor for symptoms of disease progression, medication needs, and fetal growth, increased surveillance during the prenatal period may be indicated depending on the degree of disability.
In collaboration with Physical Therapy, prescribed rehabilitation programs and stretching exercises are continued throughout pregnancy. Women with MS have increased hospital admission rates during the antepartum period.24 Even though preterm birth rates are not higher in women with MS, preterm contractions or pelvic pressure may not be perceived in women with impaired sensation.25,26 If preterm labor requires tocolysis, caution is used with administration of magnesium sulfate, which causes fatigue and weakness. When strict bed rest is prescribed, venous thromboembolism prophylaxis is recommended until the woman is able to ambulate.
Women with a history of MS-related urinary tract involvement have increased risk of infection during pregnancy. Paralysis and wheelchair dependence increases this risk. Infection of any type may cause exacerbation of the woman’s MS symptoms. Recommended routine monitoring for urinary tract infection and chronic antibiotic prophylaxis may be appropriate.26 If the woman has neurogenic bladder, increased frequency of intermittent catheterization may be needed during pregnancy to prevent incontinence. Self-catheterization may also become difficult or impossible with an enlarging uterus.26
 Known disease-specific complications, such as decreased balance, spasticity and muscle weakness, altered gait, medications (eg, hypnotics or antidepressants), reduced bone density, osteoporosis, comorbidities, dietary imbalances, and lower serum levels of vitamin D place the woman with MS at higher risk for falls.27 In addition, up to 50% of people with MS report having fallen, of which half of these falls resulted in injury.28 Pregnancy further enhances risk for falls as the woman’s body habitus changes from the enlarging uterus, and the center of gravity shifts. Assistive devices, such as a wheelchair, braces, or a walker may be utilized to decrease risk of falls. Avoiding excessive gestational weight gain, continuing with range of motion, and stretching exercises may improve ability for mobility during pregnancy.26 If the woman is in a hospital setting, an environmental assessment is recommended upon admission to improve safety. Best paths within the hospital room for mobilization, keeping the bed in the lowest position with wheels locked, and call lights placed within the woman’s reach at all times also decrease the risk of falls. Signage placed outside and/or within the woman’s room to indicate that she is at high risk for a fall is indicated according to defined hospital practices.
Known disease-specific complications, such as decreased balance, spasticity and muscle weakness, altered gait, medications (eg, hypnotics or antidepressants), reduced bone density, osteoporosis, comorbidities, dietary imbalances, and lower serum levels of vitamin D place the woman with MS at higher risk for falls.27 In addition, up to 50% of people with MS report having fallen, of which half of these falls resulted in injury.28 Pregnancy further enhances risk for falls as the woman’s body habitus changes from the enlarging uterus, and the center of gravity shifts. Assistive devices, such as a wheelchair, braces, or a walker may be utilized to decrease risk of falls. Avoiding excessive gestational weight gain, continuing with range of motion, and stretching exercises may improve ability for mobility during pregnancy.26 If the woman is in a hospital setting, an environmental assessment is recommended upon admission to improve safety. Best paths within the hospital room for mobilization, keeping the bed in the lowest position with wheels locked, and call lights placed within the woman’s reach at all times also decrease the risk of falls. Signage placed outside and/or within the woman’s room to indicate that she is at high risk for a fall is indicated according to defined hospital practices.
Dietary deficiencies are common in women with physical disabilities. This may be due to access to unhealthy foods, difficulty in chewing, dysphagia, or gastrointestinal absorption delays. In addition, iron deficiency anemia compounds physiologic anemia of pregnancy. Women prone to constipation due to limited bowel motility may resist iron supplementation.26
Antepartum medication management
Data are limited regarding treatment of MS with disease modifying agents during pregnancy. Data from studies of women with untreated disease show that approximately 25% suffer relapses during pregnancy.7,18 In a recent systematic review, common medications used with MS, interferons, glatiramer acetate, fingolimod, and natalizumab, and the effects on pregnancy outcomes were reported. Interferon-β was associated with a lower mean birth weight, shorter mean birth length, and preterm birth but did not increase the likelihood of spontaneous abortion, cesarean birth, or birth weight less than 2500 g. Glatiramer acetate was not associated with lower-birth-weight, preterm birth, congenital anomaly, or spontaneous abortion. Natalizumab exposure during pregnancy was not associated with lowerbirth-weight or gestational age at birth.29
Whereas disease-modifying medications work to reduce the disease process, none are completely effective in symptom management. Medication treatment regimes for acute relapses during pregnancy also vary because of a lack of available research and clear outcomes. If severe exacerbations of MS occur during the pregnancy, intravenous corticosteroid administration may be administered during any trimester of pregnancy and treatment is not associated with fetal malformations.20 Treatment with methylprednisolone 1 g per day, administered intravenously for 3 days may hasten the recovery but does not affect further attacks or prevent permanent disability from the attack. However, corticosteroids have potential maternal side effects of hyperglycemia, hypertension, acute psychosis, and accelerated protein catabolism leading to poor wound healing. In addition, corticosteroids cross the placental barrier and increase the risk of neonatal leukocytosis with dexamethasone use, or immunosuppression if methylprednisolone is used.3 If antepartum steroids have been utilized for symptom suppression, “stress dose” steroids may be prescribed during labor or cesarean birth. Hydrocortisone 25 mg intravenous every 8 hours in addition to the woman’s daily dose is a usual dosing regimen.30
LABOR AND BIRTH
Outcome research related to risks during labor and birth for women with MS vary, and the mechanisms of complications are not well understood. No increase in risk to the pregnancy or adverse events during labor and birth have been reported with MS.7,31 One study showed pregnant women with MS were found to have a higher incidence of labor induction, longer second stage of labor, more operative vaginal births, and cesarean deliveries, and their infants had lower birth-weights.31 However, neonatal mortality rates were not increased. Reported most recently was a 30% higher risk for cesarean birth and a 70% increase of intrauterine growth restriction as compared with healthy women.24
The determination of whether a woman should labor or undergo cesarean birth is based on obstetric reasons rather than neurologic factors.32 However, data suggest that women with physical disabilities often deliver by cesarean birth increasing the woman’s risks of anesthesia, infection, postoperative mobility, and pain management.26 Positioning options during labor and birth may be restricted because of physical limitations. With a common symptom of extreme fatigue or pelvic floor weakness, the woman may have difficulties actively pushing during the second stage of labor.32 “Labor down” techniques may be utilized to avoid fatigue and exhaustion. Operative vaginal birth should be anticipated.32
Pain management
Most published research regarding neuraxial anesthesia techniques have been reported in obstetric populations and are considered safe for women with MS. Definitive studies on pharmacologic effects of local anesthetics used in neuraxial anesthesia of women with MS are lacking.33 Local anesthetics may directly interact with MS lesions and temporarily worsen symptoms of MS by blocking sodium channels in demyelinated spinal areas. Recommendations have been made to limit neuraxial dosing to the lowest level to achieve adequate pain management in all patients during labor and birth.33 This is especially applicable in women with MS with one previous study reported a higher incidence of postpartum relapse rates in women who received greater than 0.25% bupivacaine.34 However, in a large pregnancy and MS prospective study, women who had epidural anesthesia during labor did not increase their risk of postpartum relapse or disability progression.7,35 In addition, initial studies showing a higher risk of relapse in patients who underwent spinal anesthesia have not been duplicated.33,36 Women and obstetric providers should be informed of the potential risks and benefits of each type of anesthesia to consider and plan for alternative pain management as indicated.33 Even though small reports have concerning evidence regarding spinal anesthesia, in one survey report 90% of obstetric anesthesiologist responders stated that after weighing the risks and benefits of general anesthesia, spinal anesthesia for an emergency cesarean birth would be recommended in women with MS.37 Behavioral observation scales for pain assessment is an important component of best practice for women with cognitive impairments.
POSTPARTUM
In the immediate postpartum period, extreme fatigue, pyrexia, return of sensation from neuraxial anesthesia, and/or infection may exacerbate MS symptoms or relapse. These risks increase the likelihood for falls, injury, and the inability for self-care or care of the newborn. The need for assistance during hospitalization and the immediate postpartum period should be anticipated by care providers. An increase in relapse rate in the first 3 to 6 months of postpartum is well documented in the literature but is not well understood.7,8,38
Breast-feeding
Data suggest that exclusive breast-feeding on demand produces anti-inflammatory effects because of hormonal changes. Hormone changes include higher prolactin levels and low and nonpulsatile luteinizing hormone, estradiol and progesterone levels resulting in ovarian suppression, and lactational amenorrhea.39,40 When supplemental feedings begin, prolactin levels decline, ovarian activity resumes, and menses usually returns within 2 months.
Despite evidence of the potential positive effects of breast-feeding, the fear of increased relapse risk in the postpartum period, medication use, and frequent symptoms of extreme fatigue may discourage some women from choosing to breast-feed their infant. The Pregnancy in Multiple Sclerosis data established no increased risk of postpartum relapse or disability progression in women who breast-fed their newborns.7 Two recent studies have shown potential protective effects of breast-feeding. In both studies, women who exclusively breast-fed had significantly lower disease activity during the first 3 months postpartum than those who did not breast-feed exclusively or not at all.21,41 Data from Pakpoor and colleagues41 found that women who breast-fed had almost a 50% reduction in postpartum MS relapse compared with women who did not breastfeed. However, this meta-analysis had limiting variables due to definition and measurement of breast-feeding exposure, relapse outcomes, and disease severity ratings during pregnancy.
Postpartum medication management
With a return to cellular mediated immune dominance in the postpartum period, there is a significant risk of MS symptom relapse. In women who have been previously diagnosed with MS, symptom exacerbation has been reported to increase 20% to 40% during the first 6 months following birth.7 With the knowledge of these data, the focus during the postpartum period is on prophylactic medication administration to decrease this risk.
Disease-modifying therapies and steroids can be started immediately postpartum to help cover the increased relapse risk in the first few months postpartum.42 Administration of intravenous immune globulin therapy, 0.4 g per kilogram for 5 days at 1, 6, and 12 weeks postpartum has been shown to decrease relapse rates.43 In addition to immune globulin administration, a recent study showed a significantly lower relapsing rate during the first 3 months postpartum in women who had disease-modifying therapy with glatiramer acetate and interferon-beta when compared with women who did not follow these medication regimes.21
If the woman chooses to breastfeed her infant, considerations on when to resume disease-modifying medications should be planned. Obstetric or pediatric providers may discourage breastfeeding in order for the mother to begin immediate therapy. In one study performed in the United States, only 11% of providers prescribed commonly used medications to suppress MS symptoms if the woman was breast-feeding.44 With no clear evidence on drug safety, as a general rule a woman may use FDA class A or B drugs if lactating. However, the transfer of large molecules, such as interferon-β, to the human milk is unlikely to be significant amounts to cause harm to the infant.38
Contraception and future family planning
As during the preconceptual period, family planning discussions should occur postpartum. Depending on the level of neurologic disability, potential effects of prolonged medication discontinuation should be considered if another pregnancy is planned in the near future. Hormonal contraception options are not contraindicated and have no adverse effects of incidence, overall prognosis, or disability severity in women with MS.21 In fact, one study suggests that estrogen may have protective effects against disease progression.45 However, other prescribed medications to treat symptoms of MS, such as carbamazepine used to treat muscle spasms or modafinil to treat fatigue, may inhibit the effectiveness of oral contraceptives. Because bladder infections are increased with diaphragm use, other forms of contraception may be prescribed.
Discharge planning and teaching
Cognitive impairments common in MS must be taken into consideration with discharge planning and when teaching is provided. Cognitive impairment often leads to depression, limited activity, and social withdrawal. Giving the patient written materials to take home allows the woman or family members to review instructions when needed to answer questions that may arise after discharge and to reinforce previous teaching.
Implementing family-centered care in the care of the infant and mother during hospitalization will provide opportunity for return demonstration of care and assist in assessment of home needs for the mother and infant.
Multiple sclerosis is not a fatal diagnosis though survival of patients with MS is approximately 5 to 10 years less than the general population.20 Symptoms of MS negatively affect quality of life, and suicide rates have been found to be higher in patients with MS.20 Routine postpartum depression screening may be more frequent depending on assessment findings. Social services consultation and researched community resources may be helpful for women and families as indicated, which may include transportation services for postpartum follow-up. Independent care should be encouraged, utilizing assistive devices and adaptive equipment. Care of the infant may be enhanced using cribs with side openings, lowered dressing tables, or carriers attached to the mother’s wheelchair. Removing barriers for self-care and breastfeeding may include lactation consultation, occupational therapy, and positional aids.27
CONCLUSION
Even though there has been a surge in recent research in MS in pregnancy, significant gaps in knowledge remain. Clear evidence suggests the immunosuppressive benefits of pregnancy on relapse symptoms. In addition, long-term prognosis is not affected by pregnancy. However, postpartum relapse risk factors and safety of common medications used to suppress symptoms of MS in lactating women are unclear. Providing supportive care throughout preconception, pregnancy, labor, birth, and postpartum is especially important. Table 4 summarizes previously discussed nursing care issues.
REFERENCES
1. Marchiondo K. Multiple sclerosis. Medsurg Nurs. 2010; 19(5):303–304.
2. Hauser SL, Goodin DS. Multiple sclerosis and other demyelinating diseases. In: Kasper DL, Harrison TR, eds. Harrison’s Principles of Internal Medicine. 17th ed. New York, NY: McGraw-Hill Medical Pub Division; 2008:2611–2620.
3. Houtchens MK, Lublin FD, Miller AE, Knoury SJ. Multiple sclerosis and other inflammatory demyelinating disease of the central nervous system. In: Daroff RB, Fenichel GM, Jankovic J, Mazziotta JC, eds. Bradley’s Neurology in Clinical Practice. 6th ed. Philadelphia, PA: Elsevier; 2012:1283–1313.
4. Orton S, Herrera B, Yee I, et al. Sex ratio of multiple sclerosis in Canada: a longitudinal study. Lancet Neurol. 2006;5: 932–936.
5. Ramagopalan S, Yee I, Byrnes J, Guimond C, Ebers G, Sadovnick D. Term pregnancies and the clinical characteristics of multiple sclerosis: a population-based study. J Neurosurg Psychiatry. 2012;8(83):793–795.
6. Koch M, Uyttenboogaart M, Heersema D, Steen C, de Keyser J. Parity and secondary progression in multiple sclerosis. J Neurol Neurosurg Psychiatry. 2009;80:676–678.
7. Vukusic S, Hutchinson M, Hours M, et al. Pregnancy and multiple sclerosis (The PRIMS Study): clinical predictors of post-partum relapse. Brain. 2004;127:1353–1360.
8. Confavreux C, Hutchinson M, Hours M, Cortinovis-Tourniaire P, Moreau T. Rate of pregnancy-related relapse in multiple sclerosis. Pregnancy in multiple sclerosis group. N Engl J Med. 1998;339:285–291.
9. Drew PD, Chavis JA. Female sex steroids: effects upon microglial cell activation. J Neuroimmunol. 2000;111:77–85.
10. Ponsonby AL, Lucas RM, van der Mei IA, et al. Offspring number, pregnancy, and risk of a first clinical demyelinating event: the AusImmune Study. Neurology. 2012;78:867–874.
11. Dyment DA, Ebers GC, Schubert A. Genetics of multiple sclerosis. Lancet Neurol. 2004;3(2):104–110.
12. Crabtree-Hartman E. Sex differences in MS. Continuum Lifelong Learn Neurol. 2010;16(5):193–210.
13. Grinsted L, Heltberg A, Hagen C, Djursing H. Serum sex hormone and gonadotropin concentrations in premenopausal women with multiple sclerosis. J Intern Med. 1989;226: 241–244.
14. Demirkiran M, Sarica Y, Uguz S, Yerdelen D, Aslan K. Multiple sclerosis patients with and without sexual dysfunction: are there any differences? Mult Scler. 2006;12(2):209–214.
15. Reininghaus E, Reininghaus B, Fitz W, Hecht K, Bonelli RM. Sexual behavior, body image, and partnership in chronic illness: a comparison of Huntington’s disease and multiple sclerosis. J Nerv Ment Dis. 2012;200(8):716–720.
16. Fletcher SG, Castro-Borrero W, Remington G, Treadaway K, Lemack GE, Frhman EM. Sexual dysfunction in patients with MS: a multidisciplinary approach to evaluation and management. Nat Clin Pract Urol. 2009;6(2):96–107.
17. Voskuhi RR. Assisted reproduction technology in multiple sclerosis. Giving birth to a new avenue of research in hormones and autoimmunity. Ann Neurol. 2012;72:631–632.
18. Hellwig K, Beste C, Brune N, et al. Increased MS relapse rate during assisted reproduction technique. J Neurol. 2008;255(4):592–593.
19. Foucher ML, Vukusic YS, Confavreux C, et al. Increased risk of multiple sclerosis relapse after in vitro fertilization. J Neurol Neurosurg Psychol. 2012;83:796–802.
20. Tsang B. MS: diagnosis, management and prognosis. Aust Fam Phys. 2011;40:948–955.
21. Hellwig K, Haghikia A, Rockhoff M, Gold R. Multiple sclerosis and pregnancy: experience from a nationwide database in Germany. Ther Adv Neurol Disord. 2012;5:247–253.
22. Amato MP, Portaccio E, Ghezzi A, et al. Pregnancy and fetal outcomes after interferon-β exposure in multiple sclerosis. Neurology. 2010;75:1794–1802.
23. Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69(2):292–302.
24. Kelly VM, Nelson LM, Chakravarty EF. Obstetric outcomes in women with MS and epilepsy. Neurology. 2009;73:1831– 1836.
25. van der Kop ML, Pearce MS, Dahlgren L, et al. Neonatal and delivery outcomes in women with multiple sclerosis. Ann Neurol. 2011;70(1):41–50.
26. Signore C, Spong CY, Krotoski D, Shinowara NL, Blackwell SC. Pregnancy in women with physical disabilities. Obstet Gynecol. 2011;117(4):935–947.
27. Brazelier MT, van Staa TP, Uitdehaag BMJ, et al. Risk of fractures in patients with multiple sclerosis: a population-based cohort study. Neurology. 2012;78:1967–1973.
28. Matsuda P, Shumway-Cook A, Ciol M, Bombardier C, Kartin D. Understanding falls in MS: association of mobility status, concerns about falling, and accumulated impairments. Phys Ther. 2012;92(3):407–415.
29. Lu E, Wang BW, Guiimond C, Synnes A, Sadovnick D, Tremlett H. Disease modifying drugs for multiple sclerosis in pregnancy: a systematic review. Neurology. 2012;79:1130–1135.
30. Denney JM, Porter TF, Gonik DW, eds. High Risk Pregnancy: Management Options. 4th ed. St Louis, MO: Elsevier.
31. Dahl J, Myhr KM, Daltveit AK, Hoff JM, Gilhus NE. Pregnancy, delivery, and birth outcome in women with MS. Neurology. 2005;65(12):1961–1963.
32. Houtchens M. Multiple sclerosis and pregnancy. Clin Obstet Gynecol. 2013;56(2):342–349.
33. Lirk P, Birmingham B, Hogan Q. Regional anesthesia in patients with preexisting neuropathy. Int Anesthesiol Clin. 2011;49(4):144–165.
34. Bader AM, Hunt CO, Dattta S, Naulty JS, Ostheimer GW. Anesthesia for the obstetric patient with multiple sclerosis. J Clin Anesthesiol. 1998;1(1):21–24.
35. Pasto L, Portaccio E, Ghezzi A, et al. Epidural analgesia and cesarean delivery in multiple sclerosis postpartum relapses: the Italian cohort study. BMC Neurol. 2012;12:165.
36. Vercauteren M, Heytens L. Anesthetic considerations for patients with preexisting neurological deficit: are neuraxial techniques safe? Acta Anaesthesiol Scand. 2007;51(7): 831–838.
37. Drake E, Drake M, Bird J, Russell R. Obstetric regional blocks for women with multiple sclerosis: a survey of U.K. experience. Int J Obstetr Anesth. 2006;15(2): 115–123.
38. Langer-Gould A, Beaber BE. Effects of pregnancy and breastfeeding on the multiple sclerosis disease course [published online ahead of print January 27, 2013]. Clin Immunol. doi: 10.1016/j.clim.2013.01.008.
39. Glassier A, McNeilly AS, Howie PW. Pulsatile secretion of LH in relation to the resumption of ovarian activity postpartum. Clin Endocrinol. 1984;20:415–426.
40. Glassier A, McNeilly AS, Howie PW. The prolactin response to suckling. Clin Endocrinol. 1984;21:109–116.
41. Pakpoor J, Disanto G, Lacey MV, Hellwig K, Giovannoni G, Ramagopalan SV. Breast-feeding and multiple sclerosis relapses: a meta-analysis. J Neurol. 2012;259(10): 2246–2248.
42. Tsui A, Lee MA. Multiple sclerosis and pregnancy. Curr Opin Obstetr Gynecol. 2011;23:435–439.
43. Argyriou AA, Makris N. Multiple sclerosis and reproductive risks in women. Reprod Sci. 2008;15(8):755–764.
44. Coyle PK, Christie S, Fodor P, et al. Multiple sclerosis gender issues: clinical practices of women neurologists. Mult Scler. 2004;10(5):606–612.
45. Ferrero S, Esposito F, Pretta S, Ragni N. Fetal risks related to the treatment of MS during pregnancy and breast-feeding. Expert Rev Neurother. 2006;6(12):1823–1831.

