Effect of High-Intensity Exercise on Multiple Sclerosis
Why this article interests me
In the past, healthcare providers often advised individuals living with MS to avoid exercising for fear of triggering emergence of symptoms. Experts generally agree now that, just as exercise is good for almost everyone, it is equally beneficial for people with MS as well. Exercise, however, can mean different things to different people. Does a leisurely walk around the block, for example, or a regimen of stretching provide the same benefits as a high-intensity workout? Also, if, as many of the articles in this section suggest, exercise can help to prevent or delay progression of symptoms in MS, what are the underlying mechanisms that promote well-being?
This article seeks to at the very least begin to answer these questions. The investigators studied the impact of high-intensity exercise in individuals with MS and compared their benefits to those achieved by another group of people with MS who participated in stretching exercises. The authors also put forth their theories for how exercise achieves benefits in people. Enjoy this article…and remember, always talk with your healthcare provider about your interest in exercising before beginning a program.
Who participated in the study?
The investigators recruited eighteen people with MS from the Oregon Health Sciences University MS Clinic as well as the local community. They ranged in age from 18 to 65, had been diagnosed with relapsing-remitting MS, and were fully ambulatory. Eleven of them participated in 30 minutes of aerobic exercise four times a week for eight consecutive weeks. Their target heart rate was at least 70% of their maximum heart rate (that is, 220 - age = maximum heart rate X .70: for a 40 year-old person, this would be 70% of 180, or 126 beats per minute). The other seven individuals took part in a guided static stretching program, also for 30 minutes, four times a week, for eight weeks. A third group of persons who did not have MS were recruited to serve as a control group for baseline comparison.
After eight weeks, the investigators compared participants in the high-intensity and stretching groups for:
- Changes in cardiorespiratory fitness and body composition
- Changes in cognition, fatigue, and walking performance
- Changes in biomarkers indicating brain energy
What did their results show?
As you might expect, the participants who completed eight weeks of high-intensity aerobic exercise demonstrated improvements in cardiorespiratory function and body composition (that is, reduced body fat and increased lean muscle mass). What is particularly interesting is that they also demonstrated improved mental processing speed, and reduced cognitive fatigue (that is, fatigue experienced when concentrating for extended periods of time) compared to the participants who completed eight weeks of stretching exercises.
Neither the high-intensity nor the stretching group achieved significant changes to their heart rate and blood pressure, and neither showed significant improvements in walking endurance tests.
What do their results suggest about how exercise may benefit congnitive health?
Interestingly, the authors noted differences in biomarkers associated with brain energy production between the people with MS and the control group of people without MS. These differences may provide a clue to how exercise benefits people with MS. In studies of rodents, exercise increases levels of a protein called brain derived neurotrophic factor (BDNF). BDNF helps to support the survival of existing neurons in the brain and encourage the growth of new neurons.
Most neurons form prior to birth, but parts of the adult brain retain the ability to grow new neurons. BDNF is particularly active in those areas of the brain vital to learning, memory, and higher thinking— the hippocampus, cortex, and basal forebrain. It follows then, that people who exercise should have more volume in these parts of the brain compared to those who do not.
The participants in this study did not demonstrate significant increases in these areas of their brain, but the authors wonder if longer-term programs may produce these increases in brain volume. In any case, the bottom line for readers is: even relatively sedentary people with MS can see benefits —rapid benefits — from aerobic exercise.
Original Article
Effect of High-Intensity Exercise on Multiple Sclerosis Function and Phosphorous Magnetic Resonance Spectroscopy Outcomes
Anna Orban, Bharti Garg, Manoj K. Sammi, Dennis N. Bourdette, William D. Rooney, Kerry Kuehl, and Rebecca I. Spain
Medicine & Science in Sports & Exercise
Abstract
Purpose: We determined if a high-intensity aerobic exercise program would be safe, improve expected fitness and clinical outcomes, and alter exploratory phosphorous magnetic resonance spectroscopy (31P MRS) outcomes in persons with multiple sclerosis (PwMS).
Methods: This open-label prospective pilot study compared two cohorts of ambulatory PwMS matched for age, sex and V˙O2max. Cohorts underwent 8 wk of high-intensity aerobic exercise (MS-Ex, n = 10) or guided stretching (MS-Ctr, n = 7). Aerobic exercise consisted of four 30-min sessions per week while maintaining ≥70% maximal HR. Changes in cardiorespiratory fitness, clinical outcomes, and 31P MRS of tibialis anterior (TA) muscle and brain were compared. Cross-sectional 31P MRS comparisons were made between all MS participants and a separate matched healthy control population.
Results: The MS-Ex cohort achieved target increases in V˙O2max (mean, +12.7%; P = <0.001, between-group improvement, P = 0.03). One participant was withdrawn for exercise-induced syncope. The MS-Ex cohort had within-group improvements in fat mass (−5.8%; P = 0.04), lean muscle mass (+2.6%; P = 0.02), Symbol Digit Modalities Test (+15.1%; P = 0.04), and cognitive subscore of the Modified Fatigue Impact Scale (−26%; P = 0.03), whereas only the physical subscore of the Modified Fatigue Impact Scale improved in MS-Ctr (−16.1%; P = 0.007). 31P MRS revealed significant within-group increases in MS-Ex participants in TA rate constant of phosphocreatine (PCr) recovery (+31.5%; P = 0.03) and adenosine triphosphate/PCr (+3.2%; P = 0.01), and near significant between-group increases in TA PCr recovery rate constant (P = 0.05) but no significant changes in brain 31P MRS after exercise. Cross-sectional differences existed between MS and healthy control brain PCr/inorganic phosphate (4.61 ± 0.44, 3.93 ± 0.19; P = 0.0019).
Conclusions: High-intensity aerobic exercise in PwMS improved expected cardiorespiratory and clinical outcomes but provoked one serious adverse event. The 31P MRS may serve to explore underlying mechanisms by which aerobic exercise exerts cerebral benefits.
The benefits of exercise in the general population are well known, and exercise interventions to improve general fitness and reduce symptoms in multiple sclerosis (MS) are becoming mainstream (1) Fewer trials have examined the safety, tolerability, and impacts of high-intensity aerobic exercise for people with MS (PwMS), or explored the cerebral mechanisms underlying the clinical improvements.
Although PwMS are reported to have lower baseline fitness, they generally respond as expected to aerobic exerciseinterventions (2,3). These aerobic exercise programs are typically of moderate intensity defined using the American College of Sport Medicine’s Guidelines for Exercise Testing and Prescription as 50% to 70% V˙O2max as opposed to high intensity (>70%) (4). The most frequently reported clinical benefits of aerobic exercise in MS are improving fatigue, cognition, and mobility, all of which are common, disabling, and stubbornly treatment-resistant symptoms in MS (5).
The cerebral benefits of aerobic exercise are well established. In rodents, physical activity increases levels of brain-derived neurotrophic factor and growth factors associated with improved cognition, and both preventing demyelination and promoting hippocampal neurogenesis and synaptic plasticity (6). Healthy adults show increased hippocampal volumes accompanied by improvements in memory after a 12-month aerobic exercise intervention compared with stretching (7). These changes may be associated with improved cerebral perfusion and IGF-1 levels (8). Exercise also delays onset and slows progression of cognitive impairment and brain atrophy in older adults without neurologic disease (9).
The symptomatic and neuroanatomic benefits of physical activity in MS are also evident. Cross-sectional studies in MS reveal positive associations between levels of physical activity and walking performance with fatigue, cognitive processing speed, brain volumes, and tract integrity (10–12). Longitudinal studies of aerobic exercise generally, although not invariably, demonstrate improvements in fitness and MS symptoms (3,13,14). The variations in results may be due to differences between studies in choice of exercise modality, duration, intensity, comparator group activity and outcome measures. Changes in peripheral cytokines, brain-derived neurotrophic factor, and immunological factors have also been variably noted in response to aerobic exercise in MS and other populations (8,15,16).
What are the underlying mechanisms by which aerobic exercise improves brain functional outcomes and increases volumes and tract integrity? One hypothesis is that the improved mitochondrial function in skeletal muscle resulting from aerobic exercise could also occur in the brain. Improved mitochondrial function, in turn, would reduce oxidative damage, prevent apoptosis, and have restorative properties leading to improved functional outcomes (17). Skeletal muscle mitochondrial function can be demonstrated using phosphorous MR spectroscopy (31P MRS). This technique is primarily used to measure the resynthesis of phosphocreatine (PCr) after exercise as a measure of the oxidative capacity of skeletal muscle as well as demonstrating the three adenosine triphosphate (ATP), phosphocreatine (PCr), and inorganic phosphate (Pi) peaks (18). The resynthesis of PCr, measured as a rate constant of recovery (kPCr), was shown to be lower in dorsiflexor muscles of PwMS than controls immediately after exercise (19). Increased kPCr indicates the improved ability of muscle to restore PCr/ATP ratios depleted by exercise suggesting improved mitochondrial function (18). The introduction of high field (7 T) MRI has now improved the signal to noise ratio allowing a similar evaluation of energy peaks, although not kPCr, in the brain (20). To our knowledge, these outcomes have not been explored as a potential marker of mitochondrial function in relation to an aerobic exercise protocol.
In this pilot study, we hypothesized that compared with a stretching control, a short duration, high-intensity exercise program would be well tolerated and would improve the expected outcomes of cardiorespiratory fitness, body composition, and MS symptoms of cognitive dysfunction and fatigue in PwMS. As an exploratory outcome, we hypothesized that 31P MRS would detect altered ratios of ATP, PCr, and Pi in the brain mirroring the expected changes in the of the tibialis anterior (TA) muscle in response to the exercise intervention.
Materials and Methods
Study Design
The unblended prospective pilot cohort study (NCT02263339) protocol was approved by the Institutional Review Board at Oregon Health and Science University (OHSU). Written informed consent was obtained from study participants before enrollment.
Participants
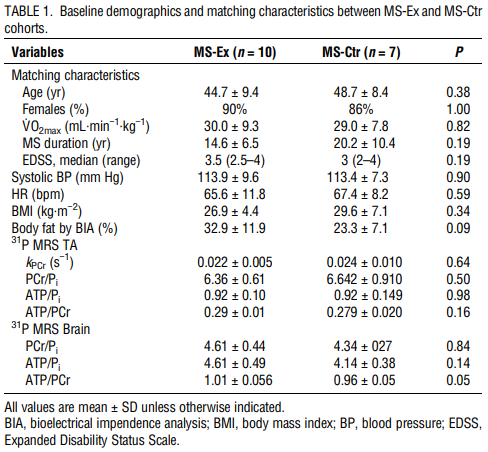
Persons with MS were recruited on a convenience basis from the OHSU MS Clinic and local community. Inclusion criteria were ages 18 to 65 yr, diagnosis of relapsing remitting MS (2010 McDonald criteria), and fully ambulatory (Expanded Disability Status Scale, ≤4.0) (21). Exclusion criteria were MS exacerbation or the use of intravenous corticosteroids or antibiotics within 30 d of screening, contraindications to MRI, or uncontrolled cardiopulmonary disease. There were no specific criteria for fatigue or cognition at baseline. MS-exercise (MS-Ex) participants were recruited first, followed by MS-control (MS-Ctr). Cohorts were matched for sex, age ±10 yr, and baseline aerobic fitness (incremental maximal exercise test, V˙O2max) ± 10%. A third cohort of healthy controls (HC) matched to the MS-Ex cohort for sex; age, ±10 yr; and baseline V˙O2max test, ±10% was recruited for cross-sectional comparison of 31P MRS outcomes.
Study Interventions
The aerobic exercise intervention consisted of four sessions per week of aerobic exercise for 30 min maintained at a target HR for eight consecutive weeks. Exercise training target HR was at least 70% of measured maximal HR for each subject. This target HR was determined by the HR during exercise testing when the RER was between 0.9 and 0.99 which is just below the anaerobic threshold. This allowed subjects to aerobically exercise train without being anaerobic. Aerobic exercise sessions were directly supervised by an exercise physiologist at the OHSU Human Performance Laboratory (HPL). Participants exercised on a treadmill (n = 9) or cycle ergometer (n = 1) chosen by the physiologist based on individual abilities. The study goal was a 5% to 15% increase in V˙O2max per expert opinion (K.K.) given expectations for a population with a chronic disease. The control intervention was a guided static stretching program for 30 min·d−1, 4 d·wk−1, for 8 wk. Initial training and a paper guide to the stretches was provided by an exercise physiologist at the HPL, followed by HPL visits every 2 wk for the same exercise physiologist to review stretching logs and encourage adherence to the program. All study participants were instructed to avoid any other change to baseline activity levels or dietary habits for the duration of the study. Reports of adverse events were collected during study visits and reviewed descriptively, and compliance with study visits was tabulated.
Study Outcomes
Cardiorespiratory fitness testing
All participants underwent measurements of resting systolic blood pressure and HR. After pulmonary function tests, participants completed a physician-supervised, electrocardiogram-monitored V˙O2max test with measurement of maximal oxygen uptake in accordance with the published guidelines (22). A maximal test was defined as one in which the participant reached a plateau in oxygen uptake, a RER above 1.1, or in which the participant stopped despite urging by the testing staff. Aerobic capacity outcomes of interest were V˙O2max (mL·kg−1·min−1), maximum work (W) and total exercise time.
Body composition testing
Participants had weight, body mass index, body fat percentage by bioelectrical impedance (Tanita BC-558 bioelectrical impedance analyzer), fat mass and lean mass tested at baseline and after the 8-wk intervention.
Phosphorus magnetic resonance spectroscopy
A Siemens 7T Magnetom system (Erlangen, Germany) was used to collect 31P MRS data before and after the study interventions. The 31P MRS TA muscle protocol used a dual-tuned 31P/1H surface coil (4 × 9.5 cm oval, with longer axis aligned to the muscle length) positioned over the center of the TA muscle of the right leg. Right legs were positioned in a home-built exercise device consisting of a Plexiglas foot pedal and adjustable rubber band that isolated dorsiflexion of the ankle against a fixed load. 31P MRS protocol of the TA muscle consisted of a 2- to 3-min period of baseline rest, a 3-min exercise period estimated to deplete phosphocreatine (PCr) by 20% to 40%, and a 4- to 10-min recovery period during which no exercise was performed. The exercise consisted of a foot-flexion exercise synchronized to a metronome set to 40 bpm 31P spectra were acquired continuously at 1.2-s intervals throughout. For brain, high-resolution MPRAGE anatomic (0.8 mm isotropic) images were acquired for tissue segmentation in a sagittal orientation (23). A three-dimensional brain 31P MRS was performed using a 31P head coil with a 1Halo coil setup (23). Low-resolution phosphorus B1 maps were acquired for RF coil inhomogeneity correction.
The primary 31P MRS TA outcome of interest was the kPCr after a bout of mild exercise. Additionally, ratios of PCr and total ATP to Pi and ratio of total ATP to PCr were captured for both TA and brain. Gamma-ATP (γ-ATP) was used as the marker of total ATP in this study.
Clinical outcomes
The Symbol Digit Modalities Test (SDMT), a measure of mental processing speed, was the cognitive measure (24). The patient-reported Modified Fatigue Impact Scale (MFIS), validated for use in MS, assessed fatigue (25). The total score consists of physical, cognitive and psychosocial subscores. The Timed Up-and-Go (TUG) test consisting of rising from a chair, walking 7 m, turning, and returning to the chair measured timed mobility, and the 6-min walk test (6MWT) measured walking endurance (26,27).
Statistical Analysis
Statistical analysis was performed using Stata 13.1 (28). Baseline demographics (Table 1) were analyzed using independent two-sample t-tests (continuous variables), Fisher Exact tests (categorical variables) and Mann-Whitney U test (ordinal variables). To determine whether there is any difference between prescore and postscore within each group (Tables 2, 3, and 4), paired t-tests were used. Pre–post change score was calculated by subtracting prescore from postscore (postscore minus prescore). Between-group change P value shows if change score (postscore minus prescore) differs between MS-Ex and MS-Control groups and was calculated using independent two-sample t-tests.
Results
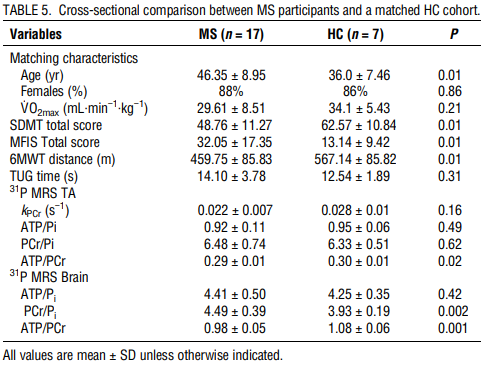
The study was conducted between November 2014, and April 2016. Eighteen MS participants consented and started the study. One MS-Ex participant was withdrawn from the study after a near-syncopal event at one visit and a syncopal event after completion of the exercise session on another visit prompting a visit to the emergency department. The participant, whose data were not included in analysis, had a childhood history of exercise-induced syncope thought to have been outgrown. There were no other adverse events. Compliance with study visits was 99.4%. At baseline, 8 of the 10 MS-Ex achieved a Max RER 1.1 or greater, whereas all MS-Ctr and HC participants met this target. At exit, only one MS-Ex participant did not meet RER 1.1. When RER was not met, the V˙O2max test was terminated by the participant. MS-Ex and MS-Ctr cohorts did not differ significantly in matching characteristics of age, sex, V˙O2max (Table 1). The HC cohort compared for baseline 31P MRS outcomes were younger (36.0 ± 7.5, 46.3 ± 8.9 yr, P = 0.01) than the MS participants (Table 5).
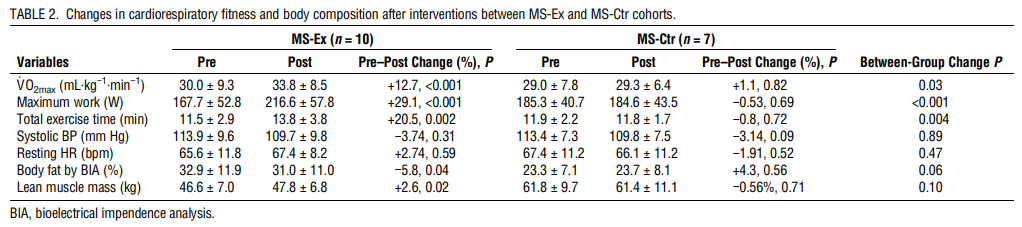
Changes in cardiorespiratory fitness and body composition
Nine of 10 MS-Ex achieved the study goal of 5% to 15% increase in V˙O2max, with mean increase of 12.7% (P < 0.001) versus none of the MS-Ctr achieving this goal (mean increase, 1.1%; P = 0.82; between group difference P = 0.03). Eight of 10 MS-Ex achieved ≥10% increase in V˙O2max, considered clinically meaningful (3). Maximum work and total exercise time also improved in the MS-Ex cohort with significant within- and between-group changes. Neither intervention group demonstrated significant changes in HR or blood pressure parameters. For body composition, percent body fat and lean muscle mass improved within the MS-Ex cohort only (−5.8%, P = 0.04, and +2.6%, P = 0.02, respectively), although neither reached a statistically significant improvement over the MS-Ctr cohort (Table 2).
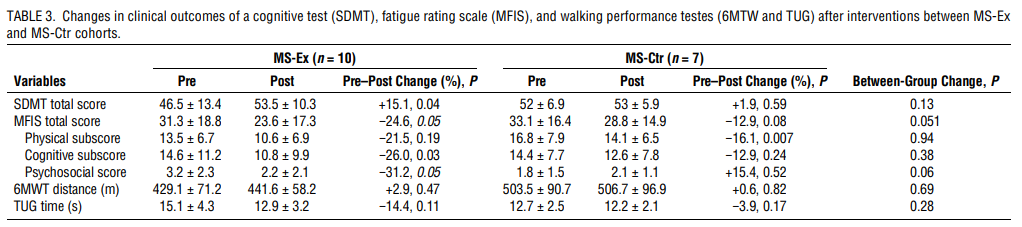
Changes in clinical outcomes of cognition, fatigue, and walking performance
The MS-Ex cohort demonstrated statistically significant improvements in mean SDMT score from baseline (mean, +7 points, +15.1%; P = 0.04), whereas MS-Ctr did not (mean, +1, +1.9%; P = 0.59; between-group P = 0.13). Similarly, the cognitive fatigue subscore of the MFIS improved significantly in the MS-Ex cohort from baseline (−26%, P = 0.03) but without a significant between group difference. Neither cohort exhibited significant changes in the TUG or 6MTW (Table 3).
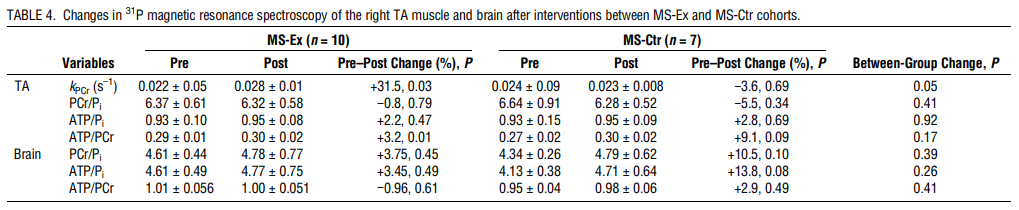
Changes in 31P MRS of the right TA and Brain
In the right TA muscle, the MS-Ex cohort demonstrated a significant within-group increase in the kPCr (+31.5%, P = 0.03) and ATP/PCr ratio (+3.2%, P = 0.01). The change in kPCr was nearly but not significantly improved over the MS-Ctr cohort (P = 0.05). No significant changes were seen in 31P MRS outcomes in the brain after interventions for either cohort (Table 4).
Discussion
In this 8-wk study, PwMS completing a high-intensity aerobic exercise program demonstrated improvements in cardiorespiratory function, body composition, mental processing speed (SDMT), and cognitive fatigue (MFIS) compared with PwMS doing a stretching program. Overall, the aerobic exercise program was well tolerated; however, the occurrence of exercise-induced near syncope in one MS-Ex subject suggests caution when initiating similar programs. Important to the study design was the matched control group stretching program of equal frequency and duration along with professional support from study staff rather than a waitlist control. Improvement in TA 31P MRS outcomes supported the V˙O2max changes found on cardiorespiratory testing. Although cerebral 31P MRS did not change after the aerobic exercise intervention, cross-sectional differences these markers of brain energy production between MS and matched HC may point to the as yet unknown underlying mechanisms by which aerobic exercise exerts its cognitive benefits.
Although there are now several well-designed studies demonstrating the safety and efficacy of high-intensity aerobic exercise in MS populations, at the time the present study was conceived, this was less clear (3) (29,30). Historically, PwMS were advised to avoid exercise for fear of worsening their disease, in part from overheating. The present study supports that even relatively sedentary PwMS can benefit rapidly from aerobic exercise. The reemergence of exercise-induced syncope in one study participant reminds us that graded intensity and/or direct supervision may be warranted in persons not accustomed to high-intensity exercise. Questions addressed in newer studies include evaluating combinations of aerobic and resistance training as well as developing exercise programs for nonambulatory PwMS to further inform clinical guidelines.
The MS-Ex participants in our study demonstrated impressive increases in V˙O2max, with 90% achieving the study goal of ≥5%, and 80% achieving what is now considered a clinically meaningful ≥10% improvement by study end (3). At the same time, the aerobic exercise participants increased their SDMT score by six points more than the MS-Ctr group, an increase that can be considered clinically meaningful (31). The improvements parallel SDMT changes found in other high-intensity aerobic exercise studies with similar V˙O2max gains (32). The MFIS is also utilized in aerobic exercise studies and appears sensitive to exercise interventions (13,32). Although not all studies find improvements in cognitive tests and fatigue self-ratings, subgroup analyses sometimes reveal that the most impaired are most likely to demonstrate a benefit, suggesting a ceiling effect of the tests (33). Our study did not select for baseline cognitive impairment or fatigue; however, our comparison with HC demonstrated baseline cross-sectional differences (Table 5). Ceiling effects may have also played a role in the lack of improvement in the 6MWT and TUG walking tests, which have been shown to improve after an exercise intervention in an MS population with a higher level of baseline disability, although baseline 6MTW results was also lower in the MS than HC participants (13). As a pilot study, sample size was not powered on these clinical outcomes which might require larger numbers, longer studies, and possibly measures not thought a priori to change after such a brief intervention, such as patient-reported psychosocial and participation outcomes (34). Overall, the clinical benefits achieved in this study were as expected based on current literature.
The mechanisms by which aerobic exercise improves cognitive function are debated and are likely many. In a recent review, El-Sayes et al. (35) proposes a model for neuroplasticity after acute and chronic effects of aerobic exercisestarting from molecular and cellular changes, and leading to structural/functional, and finally behavioral changes. This more general model of neuroplasticity appears to hold true for MS as aerobic exercise favorably changes levels of neurotrophins, neurotransmitters, inflammatory factors, hormones, neuromodulators, and more which are also associated with improved cognition, although these findings are neither specific to MS populations, nor are they detected in every study (12,15,36). Although presented sequentially, these structural and behavioral changes can occur early as evidenced by neurogenesis detected in exercising rodents within days, verbal memory and serum matrix metalloproteinases after 3 wk in MS, and increased hippocampal volume and resting-state functional connectivity after 3 months (35,37,38). Our own brain MRI segmentation results on participants in this study did not demonstrate consistent volumetric changes in hippocampal, thalamic, or other brain substructures (see Table, Supplemental Digital Content 1, Changes in deep gray matter volumes after aerobic exercise among the MS-Exercise cohort, http://links.lww.com/MSS/B509). Longer-term exercise studies do more consistently demonstrate growth of brain substructures correlating with cognitive improvement (7,39).
Demonstration of improved bioenergetic function after aerobic exercise in the TA muscle using 31P MRS raises the possibility that aerobic exercise also improves mitochondrial function in the brain. Increased oxygen consumption and glucose metabolism might, in turn, drive the molecular and cellular processes leading to neuroplasticity and improved cognition. We found cross-sectional differences in ATP metabolites between MS and HC populations, similar to Kauv et al. (20), although they expressed the peaks as percentages rather than ratios. Unclear is if the lack of change in metabolite ratios in the MS-Ex cohort after aerobic exercise was a sample size, effect size, or study duration issue, or a true finding. However, the baseline differences encourage further exploration into this potential biomarker by addressing these study design issues.
The study was limited by the small sample size and restricted permitted disability among MS participants which were due to the pilot nature of the study. Other limitations stemmed from the study design including the brevity of cognitive and fatigue testing. The SDMT is measure of mental processing speed and not specific to the cognitive deficits such as learning and memory known to commonly occur in MS. Validated MS batteries of cognitive tests exist that might have greater sensitivity to demonstrate efficacy of the interventions (40). Yet, newer evidence suggests that mental processing speed may be the cognitive domain best associated with aerobic capacity and best stand-alone cognitive task for MS (14,31). Other study limitations include lack of confounder assessments, such as mood and sleep. Finally, the lack of randomized assignment to intervention cohorts tempers the robustness of the conclusions.
Conclusions
In conclusion, a brief, high-intensity, aerobic exercise intervention compared with stretching resulted in expected improved fitness, body composition, and clinically significant increases in cognitive performance and cognitive fatigue in PwMS. The adverse event suggests caution when starting high-intensity aerobic exercise in PwMS. The cross-sectional differences in cerebral 31P MRS encourage further exploration of this potential biomarker of the bioenergetic contributions to neuroplasticity in response to aerobic exercise.
Research reported in this publication was supported by National Center for Advancing Translational Sciences of the National Institutes of Health under award number UL1TR0002369. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Funding support was provided by the National Multiple Sclerosis Society, award number CA 1073-A-4 (RS) as well as the Race to Erase MS Foundation (RS). No authors declare any conflicts of interest with companies or manufacturers who might benefit from the results of this study. The results of the present study do not constitute endorsement by ACSM. All results of the study are presented clearly, honestly, and without fabrication, falsification, or inappropriate data manipulation.
References
1. Latimer-Cheung AE, Martin Ginis KA, Hicks AL, et al. Development of evidence-informed physical activity guidelines for adults with multiple sclerosis. Arch Phys Med Rehabil. 2013;94(9):1829–36.e7.
2. Lambert CP, Archer RL, Evans WJ. Muscle strength and fatigue during isokinetic exercise in individuals with multiple sclerosis. Med Sci Sports Exerc. 2001;33(10):1613–9.
3. Platta ME, Ensari I, Motl RW, Pilutti LA. Effect of exercise training on fitness in multiple sclerosis: a meta-analysis. Arch Phys Med Rehabil. 2016;97(9):1564–72.
4. American College of Sports Medicine’s Guidelines for Exercise Testing and Prescription. Philadelphia; 2013.
5. Sandroff BM, Motl RW, Scudder MR, DeLuca J. Systematic, evidencebased review of exercise, physical activity, and physical fitness effects on cognition in persons with multiple sclerosis. Neuropsychol Rev. 2016;26(3):271–94.
6. Kim TW, Sung YH. Regular exercise promotes memory function and enhances hippocampal neuroplasticity in experimental autoimmune encephalomyelitis mice. Neuroscience. 2017;346:173–81.
7. Erickson KI, Voss MW, Prakash RS, et al. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci U S A. 2011;108(7):3017–22.
8. Maass A, Duzel S, Brigadski T, et al. Relationships of peripheral IGF-1, VEGF and BDNF levels to exercise-related changes in memory, hippocampal perfusion and volumes in older adults. NeuroImage. 2016;131:142–54.
9. Carvalho A, Rea IM, Parimon T, Cusack BJ. Physical activity and cognitive function in individuals over 60 years of age: a systematic review. Clin Interv Aging. 2014;9:661–82.
10. Sebastiao E, Hubbard EA, Klaren RE, Pilutti LA, Motl RW. Fitness and its association with fatigue in persons with multiple sclerosis. Scand J Med Sci Sports. 2017;27(12):1776–84.
11. Fritz NE, Keller J, Calabresi PA, Zackowski KM. Quantitative measures of walking and strength provide insight into brain corticospinal tract pathology in multiple sclerosis. Neuroimage Clin. 2017;14:490–8.
12. Prakash RS, Snook EM, Motl RW, Kramer AF. Aerobic fitness is associated with gray matter volume and white matter integrity in multiple sclerosis. Brain Res. 2010;1341:41–51.
13. Briken S, Gold SM, Patra S, et al. Effects of exercise on fitness and cognition in progressive MS: a randomized, controlled pilot trial. Mult Scler. 2014;20(3):382–90.
14. Langeskov-Christensen M, Eskildsen S, Stenager E, et al. Aerobic capacity is not associated with most cognitive domains in patients with multiple sclerosis—a cross-sectional investigation. J Clin Med. 2018;7(9).
15. Briken S, Rosenkranz SC, Keminer O, et al. Effects of exercise on Irisin, BDNF and IL-6 serum levels in patients with progressive multiple sclerosis. J Neuroimmunol. 2016;299:53–8.
16. Spielman LJ, Little JP, Klegeris A. Physical activity and exercise attenuate neuroinflammation in neurological diseases. Brain Res Bull. 2016;125:19–29.
17. Lassmann H, van Horssen J. Oxidative stress and its impact on neurons and glia in multiple sclerosis lesions. Biochim Biophys Acta. 2016;1862(3):506–10.
18. Argov Z, Löfberg M, Arnold DL. Insights into muscle diseases gained by phosphorus magnetic resonance spectroscopy. Muscle Nerve. 2000; 23(9):1316–34.
19. Kent-Braun JA, Sharma KR, Miller RG, Weiner MW. Postexercise phosphocreatine resynthesis is slowed in multiple sclerosis. Muscle Nerve. 1994;17(8):835–41.
20. Kauv P, Ayache SS, Creange A, et al. Adenosine triphosphate metabolism measured by phosphorus magnetic resonance spectroscopy: a potential biomarker for multiple sclerosis severity. Eur Neurol. 2017; 77(5–6):316–21.
21. Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69(2):292–302.
22. Thompson PD, Arena R, Riebe D, Pescatello LS, American College of Sports M. ACSM’s new preparticipation health screening recommendations from ACSM’s guidelines for exercise testing and prescription, ninth edition. Curr Sports Med Rep. 2013;12(4):215–7.
23. Sammi MK, Spain RI, Garg B, Kuehl K, Rooney WD. Phosphorus MR spectroscopy as a biomarker of improved tissue metabolism after aerobic exercise in multiple sclerosis at 7T [Abstract]. In: 24th Annual Meeting & Exhibition of ISMRM. Singapore, 07–13; 2016.
24. Parmenter BA, Weinstock-Guttman B, Garg N, Munschauer F, Benedict RH. Screening for cognitive impairment in multiple sclerosis using the symbol digit modalities test. Mult Scler. 2007;13(1):52–7.
25. Fisk JD, Ritvo PG, Ross L, Haase DA, Marrie TJ, Schlech WF. Measuring the functional impact of fatigue: initial validation of the fatigue impact scale. Clin Infect Dis. 1994;18(1 Suppl):S79–83.
26. Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991; 39(2):142–8.
27. Savci S, Inal-Ince D, Arikan H, et al. Six-minute walk distance as a measure of functional exercise capacity in multiple sclerosis. Disabil Rehabil. 2005;27(22):1365–71.
28. StataCorp. Stata Statistical Software: Release 13. College Station, TX: StataCorp LP; 2013.
29. Zimmer P, Bloch W, Schenk A, et al. High-intensity interval exercise improves cognitive performance and reduces matrix metalloproteinases-2 serum levels in persons with multiple sclerosis: a randomized controlled trial. Mult Scler. 2017;24:1635–44: 1352458517728342.
30. Skjerbaek AG, Naesby M, Lutzen K, et al. Endurance training is feasible in severely disabled patients with progressive multiple sclerosis. Mult Scler. 2014;20(5):627–30.
31. Benedict RH, DeLuca J, Phillips G, LaRocca N, Hudson LD, Rudick R. Validity of the symbol digit modalities test as a cognition performance outcome measure for multiple sclerosis. Mult Scler. 2017; 23(5):721–33.
32. Coghe G, Corona F, Marongiu E, et al. Fatigue, as measured using the Modified Fatigue Impact Scale, is a predictor of processing speed improvement induced by exercise in patients with multiple sclerosis: data from a randomized controlled trial. J Neurol. 2018;265(6):1328–33.
33. Mäurer M, Schuh K, Seibert S, et al. A randomized study to evaluate the effect of exercise on fatigue in people with relapsing–remitting multiple sclerosis treated with fingolimod. Mult Scler J Exp Transl Clin. 2018;4(1):2055217318756688.
34. Learmonth YC, Marshall-McKenna R, Paul L, Mattison P, Miller L. A qualitative exploration of the impact of a 12-week group exercise class for those moderately affected with multiple sclerosis. Disabil Rehabil. 2013;35(1):81–8.
35. El-Sayes J, Harasym D, Turco CV, Locke MB, Nelson AJ. Exerciseinduced neuroplasticity: a mechanistic model and prospects for promoting plasticity. Neuroscientist. 2018;1073858418771538.
36. Basso JC, Suzuki WA. The effects of acute exercise on mood, cognition, neurophysiology, and neurochemical pathways: a review. Brain Plast. 2017;2(2):127–52.
37. van Praag H, Kempermann G, Gage FH. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999;2(3):266–70.
38. Leavitt VM, Cirnigliaro C, Cohen A, et al. Aerobic exercise increases hippocampal volume and improves memory in multiple sclerosis: preliminary findings. Neurocase. 2014;20(6):695–7.
39. Rehfeld K, Luders A, Hokelmann A, et al. Dance training is superior to repetitive physical exercise in inducing brain plasticity in the elderly. PLoS One. 2018;13(7):e0196636.
40. Benedict RH, Fischer JS, Archibald CJ, et al. Minimal neuropsychological assessment of MS patients: a consensus approach. Clin Neuropsychol. 2002;16(3):381–97.


