Ocular Motor Manifestations of Multiple Sclerosis
Why is this important to me?
Eye movement disorders can occur whether your MS is acute (relapsing) or chronic (progressive or due to previous relapses). Early identification and treatment of these problems can provide a good visual outcome and improve your quality of life.
What is the objective of this study?
Eye movement disorders often show no symptoms, but over time, chronic eye muscle problems can impair vision. Several types of eye movement disorders can occur if you have MS.
Misalignment of eyes that causes double vision (called “diplopia”)
- Restricted eye movements (called “paralytic”)
- Gaze abnormality caused by impaired horizontal eye movements (called “internuclear ophthalmoplegia” or “INO”) is one of the most frequent eye movement problems in MS and can occur if you have acute or chronic disease. INO often has no symptoms and may disappear without treatment. Different variations of INO can occur.
- Other types of restricted eye movement problems can occur, depending on where the MS lesion is in the brain.
- Non-restricted (called “non-paralytic”) but misaligned eye movements
- Eye misalignment (called “strabismus”) can cause both of your eyes to be oriented inward (crossed), both of your eyes to be oriented outward, or both of your eyes to move upward in opposite directions (called “skew deviation”).
- Corticosteroids or other drugs may help improve this problem.
Eye movement instability that causes the sensation that objects in your visual field appear to move back and forth (called “oscillopsia”)
- Types of instability include:
- Repetitive uncontrolled movements of your eyes (called “nystagmus”)
- Abnormalities in visual fixation on an object (called “saccadic intrusions”)
- Abnormal accuracy of eye movements (called “saccadic dysmetria”)
- Most of these disorders occur if your MS is in the chronic phase.
- These disorders can be disruptive.
- Several treatments are available including gabapentin, memantine, clonazepam, 3,4-diaminopyridine, 4-aminopyridine, and baclofen.
Eye movement disorders that lack symptoms
- You may have an impaired ability to follow slowly moving objects (called “smooth pursuit”). Smooth pursuit occurs in about 30% of people with MS.
- You may lack a reflex that induces compensatory eye movement upon head rotation (called “vestibule-ocular reflexes” or “VOR”). VOR is impaired in about three-quarters of people with MS.
Even if you do not have specific visual symptoms, regular examination by an ophthalmologist is important to identify eye movement problems and possibly correct them, resulting in a better visual outcome in the long term.
How did the authors study this issue?
The authors reviewed various eye movement disorders that can occur in people with MS.
| SHARE: | |||||
Original Article
Ocular Motor Manifestations of Multiple Sclerosis
Elodie Nerrant, MD, Caroline Tilikete, MD, PhD
Journal of Neuro-Ophthalmology
Background: Multiple sclerosis (MS) is a demyelinating disease of the central nervous system leading to disability, especially in young patients. Acute or chronic lesions of MS within the brainstem and the cerebellum frequently result in ocular motor disorders.
Evidence Acquisition: This review encompasses the spectrum of ocular motor disorders in patients with MS emphasizing prevalence, examination findings, diagnostic features, functional consequences, classification of MS course, and management of these disturbances of ocular motility.
Results: Ocular motor manifestations of MS can occur acutely in relapse or chronically, the latter as a consequence of previous relapses or as a chronic course of the disease. The most frequent and specific acute ocular motor manifestation is uni- or bilateral internuclear ophthalmoplegia (INO). The most frequent chronic manifestations include INO and cerebellar ocular motor disorders such as gaze-evoked nystagmus, saccadic hypermetria, and lack of vestibulo-ocular reflex inhibition. The most disabling syndrome is pendular nystagmus.
Conclusions: The high prevalence of ocular motor manifestations emphasizes the importance of neuro-ophthalmological examination among patients with MS. Because chronic manifestations may cause minimal or no symptoms, a systematic investigation of the most common manifestations should be performed in daily practice. Appropriate treatment may improve visual outcome in some of these ocular motor disorders.
Multiple sclerosis (MS) is an immune-mediated disease of the central nervous system, characterized by inflammatory attacks against myelin (relapses) and by progressive axonal degeneration. Many neuro-ophthalmological manifestations are the consequence of relapses and/or the chronic disease phase of MS (1,2). Optic neuritis, as a classic example of afferent visual system involvement, is the most frequent relapse manifestation, and the most studied in the literature. Patients with MS also develop ocular motor disorders leading to diplopia or oscillopsia. The most common ocular motor abnormalities in patients with MS are internuclear ophthalmoplegia (INO), saccadic hypermetria, gaze-evoked nystagmus (GEN), and impaired vestibulo-ocular reflex (VOR) suppression (3,4). Although some disorders may be manifestations of acute MS relapses, they more frequently occur in the chronic disease phase, in which they persist over time (3,5,6). Although it may be asymptomatic, chronic ocular motor dysfunction can impair visual function and lead to poor visual outcome and reduced quality of life (3).
Our objective is not to provide an exhaustive list of ocular motor manifestations of MS, but rather to provide a systematic clinical approach with emphasis on the following questions. What are the techniques to properly examine eye movements in patients with MS? Which ocular motor manifestations suggest an initial demyelinating event in the absence of an existing MS diagnosis? How frequent are ocular motor manifestations in MS? Does the identified ocular motor abnormality result from a relapse or a chronic phase in a patient with previous diagnosis of MS? What should be the therapeutic approach?
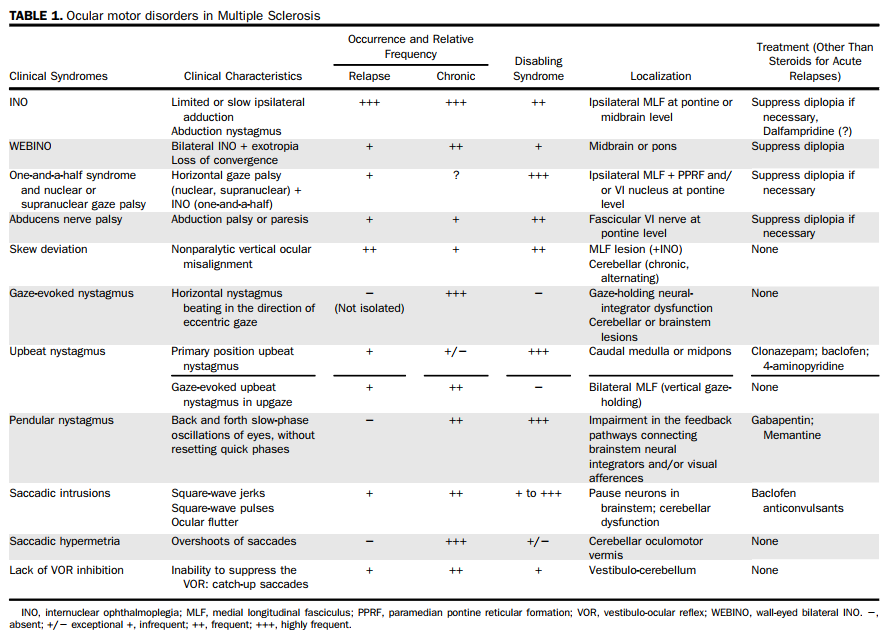
The contents of this review will be presented as follows: 1) disorders of ocular alignment and diplopia, 2) ocular instability and oscillopsia, and 3) asymptomatic ocular motor disorders. The main ocular motor disorders described are summarized in Table 1. Practical tips on appropriate bedside examination also will be described.
DISORDERS OF OCULAR ALIGNMENT AND DIPLOPIA
Paralytic
Internuclear Ophthalmoplegia
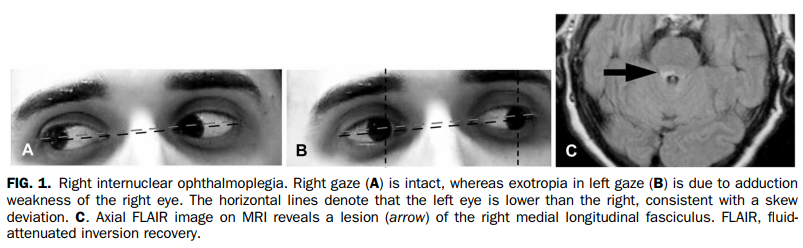
INO is one of the most frequent ocular motor disturbances in patients with MS (7). INO is characterized by ipsilateral adduction palsy and horizontal jerk nystagmus of the contralateral eye during abduction. Ipsilateral adduction should be preserved during convergence, but this dissociation may be lacking. INO is due to a lesion within the medial longitudinal fasciculus (MLF) (8), carrying adduction signal during horizontal versions from the contralateral abducens nucleus to the ipsilateral oculomotor nucleus (Fig. 1). In patients with MS, INO is frequently bilateral (9). Slowing of adducting saccades may, at times, be the only manifestation of INO. In this setting, adduction range may appear full and abducting nystagmus may be absent (10) (Practical tip E1). Recording of eye movements will increase detection of slowed adduction in INO (11).
The MLF also carries vertical, torsional, and velocity eye movement information. Thus, unilateral INO can be associated with skew deviation (with the eye contralateral to the MLF lesion being lower), whereas bilateral INO is associated with gaze-evoked upbeat nystagmus in upgaze (1,11).
INO in MS can occur either as an acute symptom of an MS attack, or as a chronic condition (Table 1). In the latter case, this results from either incomplete recovery of previous attack or from the chronic phase of the disease. Typically, patients with chronic INO are asymptomatic or have nonspecific visual complaints. In MS, unilateral INO is observed in 30% of patients, and bilateral INO in 12% (12,13), rising to 56% (31% if bilateral) in patients with persistent visual complaints (3). Although there are no data regarding the frequency of acute INO in patients with MS, MS is the cause in 32%–34% of cases of INO, 27%–28% if unilateral, and 17%–41% if bilateral (9,14). After at least 9 months, up to 60% of INO cases in patients with MS may recover (9). MLF lesions generally are very small and may, or may not, be seen on MRI. T2 sequences are best to detect MLF involvement and are superior to fluid-attenuated inversion recovery images (8).
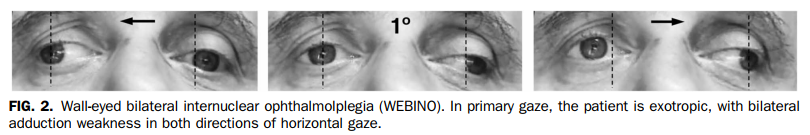
WEBINO, the acronym for wall-eyed bilateral INO, encompasses bilateral adduction deficits and bilateral abduction nystagmus (Fig. 2). The basis for exotropia, the cardinal sign of WEBINO syndrome is unknown, as it appears not be caused by loss of vergence or involvement of the medial rectus subnuclei of the third nerve nuclear complex (15,16). WEBINO may occur as a relapse or in the chronic phase of MS.
Other Brainstem Disorders
The one-and-a-half syndrome consists of an INO and an ipsilateral horizontal gaze palsy. The only remaining horizontal eye movement is abduction contralateral to the lesion. Adduction with convergence is preserved. The one-and-a-half syndrome is secondary to a lesion of the MLF and either the ipsilateral abducens nucleus (nuclear one-and-a-half) or the paramedian pontine reticular formation (PPRF) (supranuclear one-and-a-half syndrome). In nuclear syndrome, all eye movements are impaired; whereas in supranuclear dysfunction, limited voluntary rapid eye movements (saccades) can be overcome by oculocephalic stimulations (Practical tip E2). If there is ipsilateral peripheral facial nerve involvement, the clinical presentation is designated as the eight-and-a-half syndrome. Approximately 30% of cases of one-and-a-half syndrome are due to MS, mainly from acute relapses (17,18) (Table 1). In many cases, patients partially recover from a one-and-a-half syndrome, and are left with a residual INO.
Uni- or bilateral horizontal nuclear or supranuclear gaze palsies (without INO) have been reported in patients with MS (19,20). Rarely, dorsal midbrain syndrome has also been documented (21).
Ocular Motor Cranial Nerve Palsy
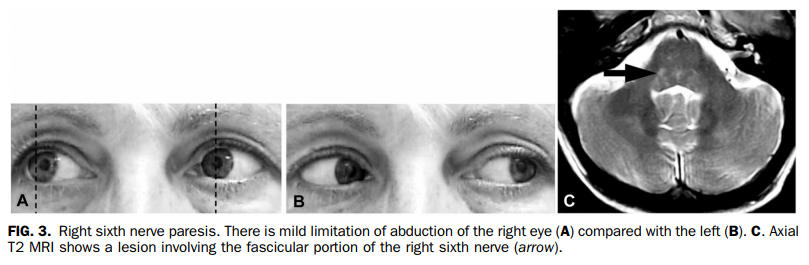
Ocular motor cranial nerve palsies in patients with MS are due to demyelination of the fascicular portion of the cranial nerves within the brainstem. Typically, these result from clinical relapses. Most often the sixth nerve is involved, but occurs during a relapse in only 0.4%–1% of patients with MS (Table 1) (Fig. 3) (22–24). Whether MS represents a frequent etiology of sixth nerve palsy is controversial; it is reported to range from 1% (24) to 12% (25,26) of causes of the sixth nerve palsy. Third nerve palsy as the presenting sign of MS occurs in 2.8% of cases (27). Partial involvement is typical with or without pupil involvement (17,28). The fourth nerve is rarely affected in MS (29).
Nonparalytic
In cases of nonparalytic strabismus, range of eye movements appears full. Demonstration of small amounts of ocular misalignment requires careful assessment of eye position using the cover-uncover and alternate cover tests (Practical tip E3).
Skew deviation is a nonparalytic vertical ocular misalignment observed in all directions of gaze (30). It is typically caused by lesions of central graviceptive vestibular pathways, including the cerebellum. Description of skew deviation is conventionally given by the side of the lower eye. With a lesion below the decussation of the vestibular pathways at the pontine level, the lower eye is on the opposite side of the lesion. In MS, acute skew deviation is frequently observed in association with contralateral INO (Table 1) (31). In the chronic phase, skew deviation reflects cerebellar involvement and may be seen as an alternating skew deviation (32).
The cerebellum also has a role in the control of the alignment of the eyes, and esophoria or esotropia (nonparalytic strabismus) occurs in chronic cerebellar disorders (33). These chronic cerebellar ocular misalignments are not specific for MS, but may suggest a chronic disease phase in patients with known MS.
Therapeutic Management of Ocular Motor Palsies or Misalignments
During attacks, corticosteroids remain essential to improve clinical outcomes, although patients may need to temporarily use ocular occlusion. Of interest, dalfampridine, a potassium channel blocker prescribed for gait impairment in MS, has led to improved adduction in INO in patients with MS (34). In some very rare cases, prism glasses, botulinum toxin, or eye muscle surgery may be warranted (9,35–37).
OCULAR INSTABILITY AND OSCILLOPSIA
Ocular instability in MS may be due to nystagmus, saccadic intrusions, or saccadic hypermetria (Practical tip E4). Those occurring most frequently in MS are GEN and saccadic hypermetria, but the most disabling disorder is pendular nystagmus (3,39). With the exception of upbeat nystagmus, which occurs in acute attacks, the other types of ocular instability reflect a chronic disease phase.
Nystagmus
Jerk Nystagmus
Different forms of jerk nystagmus have been reported in patients with MS; by far the most frequent being GEN, followed by primary position upbeat nystagmus.
GEN has been described in 16% of patients with MS (12) and up to 44% of patients with MS complaining of visual dysfunction (3). With GEN, the fast phase is in the direction of eccentric gaze, horizontal in lateral gaze, and/or vertical in upgaze. GEN is the result of a gaze-holding neural integrator dysfunction (velocity-to-position integrator). In horizontal gaze, this neural network involves the prepositus hypoglossi and the medial vestibular nuclei. Superior vestibular nuclei/MLF/midbrain interstitial nucleus of Cajal integrate ocular motor parameters in vertical gaze (16). Cerebellar flocculus and paraflocculus are connected to the brainstem network, and a feedback mechanism controls their function. Therefore, GEN may be caused by brainstem and/or cerebellar lesions. Patients with GEN are rarely symptomatic from this eye movement disorder, and it usually represents chronic disease (Table 1). Gaze-evoked upbeat nystagmus in upgaze in patients with MS may occur in association with bilateral INO, either as a result of acute relapse or as a chronic manifestation.
Primary position upbeat nystagmus is the result of a discrete lesion located in caudal medullary or pontine tegmentum level (30–43). This nystagmus can change with head position, that is, dampen or reverse in the prone or supine position.
Downbeat nystagmus (DBN) rarely has been reported in MS (44,45). It is due to bilateral flocculus impairment resulting in vestibular nuclei disinhibition (40). In MS, DBN reflects a chronic disease phase. Other kinds of jerk nystagmus have been described in MS, including periodic alternating nystagmus (46) and nystagmus associated to central vestibular disorders (47).
Pendular Nystagmus
Acquired pendular nystagmus (APN) is rare and defined by a regular back and forth slow-phase oscillation of the eyes. APN in patients with MS is characterized by small amplitude, high frequency, and great regularity (38). APN may be horizontal, vertical, or torsional, most often bilateral, and can be conjugate or dysconjugate. The persistence of APN in primary gaze results in disabling oscillopsia. At times, it is only triggered by convergence (48). It has always been described as permanent, mainly in chronic disease phase (Table 1). Mechanisms postulated for causing pendular nystagmus in MS include abnormal delays in the ocular motor feedback loops due to demyelination. This abnormal feedback loop could involve the velocity-to-position neural integrators, which normally stabilize gaze (49) and/or an abnormal visual feedback due to optic nerve pathology (50,51). Indeed, this nystagmus often is associated with chronic optic neuropathy and/or to diffuse pontine tegmental lesions. APN has been described in 11% of 284 patients with MS referred to an ophthalmologic department (52) and up to 20% in a cohort of 73 patients with MS complaining of persistent visual impairment (3). One publication has reported dampening of APN in a patient with MS with vibration of the skull using a hand held muscle massager (53).
See-saw nystagmus is characterized by supraduction and incyclotorsion of one eye, and infraduction and excycolotorsion of the other eye. A parasellar, chiasmal, or midbrain lesion often is identified, but occasionally see-saw nystagmus has been reported in patients with MS (54,55).
Saccadic Intrusions and Hypermetria
Saccadic Intrusions
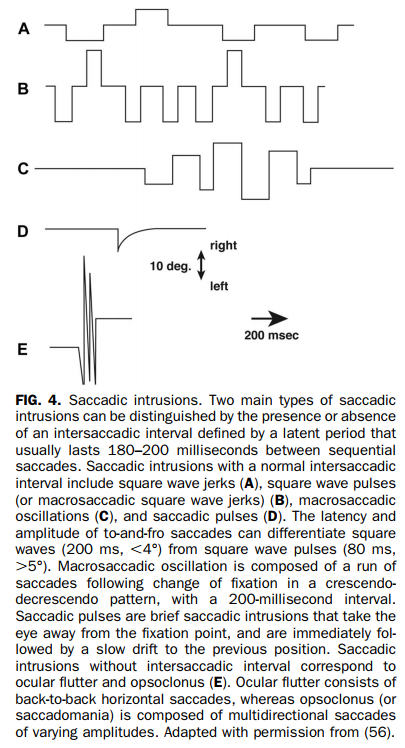
The most common saccadic intrusions in MS are square-wave jerks and macrosaccadic square-wave jerks, both occurring in association with cerebellar syndrome (56) (Fig. 4). Ocular flutter (horizontal) and opsoclonus (multidirectional) are commonly part of a paraneoplastic syndrome or following viral infections, but occasionally are caused by MS (44). These saccadic oscillations can occur either in relapse (17,57,58) or in chronic forms for ocular flutter.
Saccadic Dysmetria
Saccadic dysmetria is one of the most common chronic ocular movement abnormalities in MS, primarily as saccadic hypermetria and directional dysmetria (Practical tip E5) (7). Lesions of the dorsal oculomotor vermis and the fastigial oculomotor region are responsible for abnormal accuracy and trajectory of saccades (60,61), and observed only in chronic cerebellar syndrome (Table 1). Saccadic dysmetria is present in approximately 30%–40% of patients with MS (3,12).
Therapeutic Management of Nystagmus and Other Ocular Instability
Because nystagmus and other disorders of ocular stability are very disabling in patients with MS, a variety of treatments have been used (Table 1). Gabapentin (1,200–2,400 mg/d) and memantine (20–40 mg/d) are effective in the treatment of APN leading to an improved visual outcome (59–61). However, memantine should be used cautiously as it has been associated with a risk of worsening MS (62). Clonazepam (1.5–3.0 mg/d) and the potassium channel antagonists 3,4-diaminopyridine (40–60 mg/d) and 4-aminopyridine (10–15 mg/d) have demonstrated significant improvement of DBN (63–65). Periodic alternating nystagmus may be treated with baclofen (15–30 mg/d) (66). Saccadic intrusions may improve with baclofen or anticonvulsants (67).
ASYMPTOMATIC OCULAR MOTOR DISORDERS
Examination of eye movements, with attention to dynamic properties of saccades, smooth pursuit, and visual inhibition of VOR, may provide valuable information in detecting chronic brainstem and cerebellar disorders. Important examples include adduction saccade slowing in INO and saccadic hypermetria. An examination of patients with MS should include evaluation of smooth pursuit (See Supplemental Digital Content, Video 1, http://links.lww.com/WNO/A221). Jerk or saccadic pursuit in a nonspecific finding as it may be found in any individual who is tired, visually handicapped, or uncooperative and in patients taking sedative drugs (44). The smooth pursuit network is large and its impairment is not topographically specific, limiting the usefulness of its testing. Examination of VOR inhibition by fixation can be more valuable to detect dysfunction of the smooth pursuit/fixation system (See Supplemental Digital Content, Video 2, http://links.lww.com/WNO/A222).
Smooth pursuit has been found to be chronically impaired in 30% of patients with MS (4), and lack of VOR inhibition is reported in 70%–75% of patients with MS (3,68). Although impaired smooth pursuit may be due to cerebellar, brainstem, and hemispheric lesions, lack of VOR inhibition seems to be more specific of chronic cerebellar involvement, related to diffuse lesions of vestibulo-cerebellum (Table 1) (69).
CONCLUSION
Patients with MS may develop a broad range of efferent visual disorders due to brainstem and cerebellar dysfunction. Visual symptoms of efferent system disorders may occur at onset of disease, during relapse, or with the chronic course of MS. Moreover, patients may not notice any visual difficulty, or may report nonspecific visual complains and a systematic clinical examination is essential to recognize these ocular motor disorders. Improving ocular motor diagnosis ensures the best clinical evaluation of patients with MS to classify them, score the handicap, and propose effective treatments to limit visual disability.
PRACTICAL TIPS
Practical Tip E1: How to Examine for an Internuclear Ophthalmoplegia?
INO in MS is more frequently characterized by slowing of the adducting eye rather than adduction palsy of the ipsilateral eye. Amplitude of adduction is then maintained during slow eye movements such as smooth pursuit. Therefore, saccades should be examined by asking the patient to fixate alternatively 2 peripheral targets: a slower adduction saccade relative to a normal abduction one is easy to identify. INO in MS can be chronic, asymptomatic, and resulting either from incomplete recovery of attack or as manifestation of chronic phase. A specific search for INO as described above should be part of systematic ocular motor examination in suspected or known patients with MS.
Practical Tip E2: How to Differentiate Nuclear From Supranuclear Horizontal or Vertical Gaze Palsy?
When a patient presents with a horizontal or vertical gaze palsy, the examiner must identify if the gaze palsy is due to nuclear or supranuclear (structures that trigger saccades) dysfunction. In horizontal gaze palsy, abducens nucleus syndrome should be distinguished from PPRF syndrome. In vertical gaze palsy, oculomotor nucleus syndrome should be distinguished from dorsal midbrain syndrome, which reflects the involvement of structures that trigger vertical saccades. The oculocephalic reflex plays a key role. To evaluate the oculocephalic response, the examiner has to swiftly rotate the patient's head horizontally then vertically while the patient is fixating a stationary target such as the examiner's nose. The input that triggers the stabilizing eye movements comes either from the VOR, the visual system, and/or the neck proprioceptive afferences. Limited voluntary rapid eye movements (saccades) can be overcome by oculocephalic stimulations, suggesting a supranuclear (or nonorganic) origin.
Practical Tip E3: How to Diagnose an Ocular Misalignment?
Cover tests are helpful to demonstrate a small ocular misalignment. These tests are based on the principle that if the visual axis is not aligned with the target, a rapid eye movement (saccade) will be performed to foveate the presented target. The detection and the amplitude of this corrective saccade indicate the misalignment of the visual axis to the clinician. The patient also can verbally describe the change in the position of the target, from one covering condition to the other. Using a translucent occluder may be useful for the examiner to observe the movement of the covered eye.
The cover-uncover test reveals heterotropia or ocular deviation with binocular fixation. If the cover-uncover test does not demonstrate ocular misalignment (eyes are in orthotropic position), the examiner should proceed to the cross-cover test to demonstrate heterophoria. With both testing techniques, esotropia(phoria) is denoted by an outward movement, exotropia(phoria) by an inward movement, hypertropia(phoria) by a downward movement, and an hypotropia(phoria) by an upward movement of the ipsilateral eye.
Practical Tip E4: How to Examine for Ocular Instability?
The patient initially should be examined with eyes in the primary position of gaze, while looking at a small object located in front of him/her, with binocular then monocular viewing. Next, ocular stability have to be looked for when the patient fixates 20° to the right, to the left, up and down, and in convergence. Looking at the pupil helps to identify horizontal or vertical instability, and at conjunctival vessels for instability in the torsional plane. Focus on eyelids may detect small amplitude vertical ocular instability. To identify tiny ocular instability, optic disk stability should be checked through the ophthalmoscope or using magnification such as Frenzel goggles. Nystagmus present in primary position of gaze is always pathological. GEN is specifically triggered in eccentric gaze and consists of centrifugal slow phases followed by centripetal fast phases. GEN has to be differentiated from end-point nystagmus seen in extreme lateral gaze (30–40°) in normal individuals as a consequence of ocular fatigue with prolonged lateral gaze. Saccadic intrusions such as square wave jerks can be seen in normal individuals, particularly in the elderly.
Practical Tip E5: How to Look for Saccadic Dysmetria?
Saccades should be examined by asking the patient to fixate a central target (i.e., the examiner's nose) then a peripheral target (i.e., the examiner's finger or the tip of a pen) and back again to the central target. Saccades in each direction should be checked. This bedside examination allows the analysis of velocity, initiation (latency), accuracy, and conjugacy of saccadic eye movements. Saccades can be inaccurate, either hypometric or hypermetric, leading to forward or backward correction, respectively. This corrective saccade is then detected clinically. A directional dysmetria may also be found, such as a vertical movement in horizontal saccade. Saccadic hypometria is a frequent observation in normal subjects. In MS, saccadic dysmetria can be either saccadic hypermetria or directional dysmetria.
STATEMENT OF AUTHORSHIP
Category 1: a. Conception and design: E. Nerrant and C. Tilikete; b. Acquisition of data: E. Nerrant and C. Tilikete; c. Analysis and interpretation of data: E. Nerrant and C. Tilikete. Category 2: a. Drafting the manuscript: E. Nerrant and C. Tilikete; b. Revising it for intellectual content: C. Tilikete. Category 3: a. Final approval of the completed manuscript: E. Nerrant and C. Tilikete.
REFERENCES
1. Frohman EM, Frohman TC, Zee DS, McColl R, Galetta S. The neuro-ophthalmology of multiple sclerosis. Lancet Neurol. 2005;4:111–121.
2. Prasad S, Galetta SL. Eye movement abnormalities in multiple sclerosis. Neurol Clin. 2010;28:641–655.
3. Jasse L, Vukusic S, Durand-Dubief F, Vartin C, Piras C, Bernard M, Pélisson D, Confavreux C, Vighetto A, Tilikete C. Persistent visual impairment in multiple sclerosis: prevalence, mechanisms and resulting disability. Mult Scler. 2013;19:1618–1626.
4. Niestroy A, Rucker JC, Leigh RJ. Neuro-ophthalmologic aspects of multiple sclerosis: using eye movements as a clinical and experimental tool. Clin Ophthalmol. 2007;1:267–272.
5. Tilikete C, Jasse L, Vukusic S, Durand-Dubief F, Vardanian C, Pelisson D, Vighetto A. Persistent ocular motor manifestations and related visual consequences in multiple sclerosis. Ann N Y Acad Sci. 2011;1233:327–334.
6. Servillo G, Renard D, Taieb G, Labauge P, Bastide S, Zorzon M, Castelnovo G. Bedside tested ocular motor disorders in multiple sclerosis patients. Mult Scler. 2014;2014:732329.
7. Serra A, Derwenskus J, Downey DL, Leigh RJ. Role of eye movement examination and subjective visual vertical in clinical evaluation of multiple sclerosis. J Neurol. 2003;250:569–575.
8. Frohman EM, Zhang H, Kramer PD, Fleckenstein J, Hawker K, Racke MK, Frohman TC. MRI characteristics of the MLF in MS patients with chronic internuclear ophthalmoparesis. Neurology. 2001;57:762–768.
9. Bolanos I, Lozano D, Cantu C. Internuclear ophthalmoplegia: causes and long-term follow-up in 65 patients. Acta Neurol Scand. 2004;110:161–165.
10. Frohman TC, Frohman EM, O’Suilleabhain P, Salter A, Dewey RB Jr, Hogan N, Galetta S, Lee AG, Straumann D, Noseworthy J, Zee D, Corbett J, Corboy J, Rivera VM, Kramer PD. Accuracy of clinical detection of INO in MS: corroboration with quantitative infrared oculography. Neurology. 2003;61:848–850.
11. Ranalli PJ, Sharpe JA. Vertical vestibulo-ocular reflex, smooth pursuit and eye-head tracking dysfunction in internuclear ophthalmoplegia. Brain. 1988;111:1299–1317.
12. Downey DL, Stahl JS, Bhidayasiri R, Derwenskus J, Adams NL, Ruff RL, Leigh RJ. Saccadic and vestibular abnormalities in multiple sclerosis: sensitive clinical signs of brainstem and cerebellar involvement. Ann N Y Acad Sci. 2002;956:438– 440.
13. Meienberg O, Muri R, Rabineau PA. Clinical and oculographic examinations of saccadic eye movements in the diagnosis of multiple sclerosis. Arch Neurol. 1986;43:438–443.
14. Keane JR. Internuclear ophthalmoplegia: unusual causes in 114 of 410 patients. Arch Neurol. 2005;62:714–717.
15. Chen CM, Lin SH. Wall-eyed bilateral internuclear ophthalmoplegia from lesions at different levels in the brainstem. J Neuroophthalmol. 2007;27:9–15.
16. Leigh RJ, Zee DS. The Neurology of Eye Movement. 5th edition. Oxford, United Kingdom: Oxford University Press, 2015.
17. de Seze J, Vukusic S, Viallet-Marcel M, Tilikete C, Zephir H, Delalande S, Stojkovic T, Defoort-Dhellemmes S, Confavreux C, Vermersch P. Unusual ocular motor findings in multiple sclerosis. J Neurol Sci. 2006;243:91–95.
18. Wall M, Wray SH. The one-and-a-half syndrome–a unilateral disorder of the pontine tegmentum: a study of 20 cases and review of the literature. Neurology. 1983;33:971–980.
19. Kipfer S, Crook DW. Isolated bilateral horizontal gaze palsy as first manifestation of multiple sclerosis. Mult Scler. 2014;20:754–755.
20. Milea D, Napolitano M, Dechy H, Le Hoang P, Delattre JY, Pierrot-Deseilligny C. Complete bilateral horizontal gaze paralysis disclosing multiple sclerosis. J Neurol Neurosurg Psychiatry. 2001;70:252–255.
21. Quint DJ, Cornblath WT, Trobe JD. Multiple sclerosis presenting as Parinaud syndrome. AJNR. 1993;14:1200–1202.
22. Thomke F, Lensch E, Ringel K, Hopf HC. Isolated cranial nerve palsies in multiple sclerosis. J Neurol Neurosurg Psychiatry. 1997;63:682–685.
23. Zadro I, Barun B, Habek M, Brinar VV. Isolated cranial nerve palsies in multiple sclerosis. Clin Neurol Neurosurg. 2008;110:886–888.
24. Barr D, Kupersmith MJ, Turbin R, Bose S, Roth R. Isolated sixth nerve palsy: an uncommon presenting sign of multiple sclerosis. J Neurol. 2000;247:701–704.
25. Moster ML, Savino PJ, Sergott RC, Bosley TM, Schatz NJ. Isolated sixth-nerve palsies in younger adults. Arch Ophthalmol. 1984;102:1328–1330.
26. Peters GB III, Bakri SJ, Krohel GB. Cause and prognosis of nontraumatic sixth nerve palsies in young adults. Ophthalmology. 2002;109:1925–1938.
27. Rush JA, Younge BR. Paralysis of cranial nerves III, IV, and VI. Cause and prognosis in 1,000 cases. Arch Ophthalmol. 1981;99:76–79.
28. Seery LS, Hurliman E, Erie JC, Leavitt JA. Bilateral pupil-sparing third nerve palsies as the presenting sign of multiple sclerosis. J Neuroophthalmol. 2011;31:241–243.
29. Jacobson DM, Moster ML, Eggenberger ER, Galetta SL, Liu GT. Isolated trochlear nerve palsy in patients with multiple sclerosis. Neurology. 1999;53:877–879.
30. Brandt T, Dieterich M. Skew deviation with ocular torsion: a vestibular brainstem sign of topographic diagnostic value. Ann Neurol. 1993;33:528–534.
31. Zwergal A, Cnyrim C, Arbusow V, Glaser M, Fesl G, Brandt T, Strupp M. Unilateral INO is associated with ocular tilt reaction in pontomesencephalic lesions: INO plus. Neurology. 2008;71:590–593.
32. Keane JR. Alternating skew deviation: 47 patients. Neurology. 1985;35:725–728.
33. Versino M, Hurko O, Zee DS. Disorders of binocular control of eye movements in patients with cerebellar dysfunction. Brain. 1996;119:1933–1950.
34. Serra A, Skelly MM, Jacobs JB, Walker MF, Cohen JA. Improvement of internuclear ophthalmoparesis in multiple sclerosis with dalfampridine. Neurology. 2014;83:192–194.
35. Adams WE, Leavitt JA, Holmes JM. Strabismus surgery for internuclear ophthalmoplegia with exotropia in multiple sclerosis. J AAPOS. 2009;13:13–15.
36. Murthy R, Dawson E, Khan S, Adams GG, Lee J. Botulinum toxin in the management of internuclear ophthalmoplegia. J AAPOS. 2007;11:456–459.
37. Roper-Hall G, Cruz OA, Chung SM. Results of extraocular muscle surgery in WEBINO bilateral internuclear ophthalmoplegia patients. J AAPOS. 2008;12:277–281.
38. Tilikete C, Jasse L, Pelisson D, Vukusic S, Durand-Dubief F, Urquizar C, Vighetto A. Acquired pendular nystagmus in multiple sclerosis and oculopalatal tremor. Neurology. 2011;76:1650–1657.
39. Kim JA, Jeong IH, Lim YM, Kim KK. Primary position upbeat nystagmus during an acute attack of multiple sclerosis. J Clin Neurol. 2014;10:37–41.
40. Pierrot-Deseilligny C, Milea D. Vertical nystagmus: clinical facts and hypotheses. Brain. 2005;128:1237–1246.
41. Pierrot-Deseilligny C, Milea D, Sirmai J, Papeix C, RivaudPechoux S. Upbeat nystagmus due to a small pontine lesion: evidence for the existence of a crossing ventral tegmental tract. Eur Neurol. 2005;54:186–190.
42. Pierrot-Deseilligny C, Richeh W, Bolgert F. Upbeat nystagmus due to a caudal medullary lesion and influenced by gravity. J Neurol. 2007;254:120–121.
43. Tilikete C, Milea D, Pierrot-Deseilligny C. Upbeat nystagmus from a demyelinating lesion in the caudal pons. J Neuroophthalmol. 2008;28:202–206.
44. Barnes D, McDonald WI. The ocular manifestations of multiple sclerosis. 2. Abnormalities of eye movements. J Neurol Neurosurg Psychiatry. 1992;55:863–868.
45. Bronstein AM, Miller DH, Rudge P, Kendall BE. Down beating nystagmus: magnetic resonance imaging and neuro-otological findings. J Neurol Sci. 1987;81:173–184.
46. Matsumoto S, Ohyagi Y, Inoue I, Oishi A, Goto H, Nakagawa T, Yamada T, Kira J. Periodic alternating nystagmus in a patient with MS. Neurology. 2001;56:276–277.
47. Pula JH, Newman-Toker DE, Kattah JC. Multiple sclerosis as a cause of the acute vestibular syndrome. J Neurol. 2013;260:1649–1654.
48. Sharpe JA, Hoyt WF, Rosenberg MA. Convergence-evoked nystagmus. Congenital and acquired forms. Arch Neurol. 1975;32:191–194.
49. Das VE, Oruganti P, Kramer PD, Leigh RJ. Experimental tests of a neural-network model for ocular oscillations caused by disease of central myelin. Exp Brain Res. 2000;133:189– 197.
50. Averbuch-Heller L, Zivotofsky AZ, Das VE, DiScenna AO, Leigh RJ. Investigations of the pathogenesis of acquired pendular nystagmus. Brain. 1995;118:369–378.
51. Barton JJ, Cox TA. Acquired pendular nystagmus in multiple sclerosis: clinical observations and the role of optic neuropathy. J Neurol Neurosurg Psychiatry. 1993;5:262– 267.
52. Roodhooft JM. Summary of eye examinations of 284 patients with multiple sclerosis. Int J MS Care. 2012;14:31–38.
53. Beh SC, Tehrani AS, Kheradmand A, Zee DS. Damping of monocular pendular nystagmus with vibration in a patient with multiple sclerosis. Neurology. 2014;82:1380–1381.
54. Samkoff LM, Smith CR. See-saw nystagmus in a patient with clinically definite MS. Eur Neurol. 1994;34:228–229.
55. Sandramouli S, Benamer HT, Mantle M, Chavan R. See-saw nystagmus as the presenting sign in multiple sclerosis. J Neuroophthalmol. 2005;25:56–57.
56. Lemos J, Eggenberger E. Saccadic intrusions: review and update. Curr Opin Neurol. 2013;26:59–66.
57. Francis DA, Heron JR. Ocular flutter in suspected multiple sclerosis: a presenting paroxysmal manifestation. Postgrad Med J. 1985;61:333–334.
58. Schon F, Hodgson TL, Mort D, Kennard C. Ocular flutter associated with a localized lesion in the paramedian pontine reticular formation. Ann Neurol. 2001;50:413– 416.
59. Averbuch-Heller L, Tusa RJ, Fuhry L, Rottach KG, Ganser GL, Heide W, Büttner U, Leigh RJ. A double-blind controlled study of gabapentin and baclofen as treatment for acquired nystagmus. Ann Neurol. 1997;41:818–825.
60. Starck M, Albrecht H, Pollmann W, Dieterich M, Straube A. Acquired pendular nystagmus in multiple sclerosis: an examiner-blind cross-over treatment study of memantine and gabapentin. J Neurol. 2010;257:322–327.
61. Starck M, Albrecht H, Pollmann W, Straube A, Dieterich M. Drug therapy for acquired pendular nystagmus in multiple sclerosis. J Neurol. 1997;244:9–16.
62. Villoslada P, Arrondo G, Sepulcre J, Alegre M, Artieda J. Memantine induces reversible neurologic impairment in patients with MS. Neurology. 2009;72:1630–1633.
63. Kalla R, Glasauer S, Schautzer F, Lehnen N, Buttner U, Strupp M, Brandt T. 4-aminopyridine improves downbeat nystagmus, smooth pursuit, and VOR gain. Neurology. 2004;62:1228–1229.
64. Strupp M, Schuler O, Krafczyk S, Jahn K, Schautzer F, Buttner U, Strupp M, Brandt T. Treatment of downbeat nystagmus with 3,4-diaminopyridine: a placebo-controlled study. Neurology. 2003;61:165–170.
65. Young YH, Huang TW. Role of clonazepam in the treatment of idiopathic downbeat nystagmus. Laryngoscope. 2001;111:1490–1493.
66. Halmagyi GM, Rudge P, Gresty MA, Leigh RJ, Zee DS. Treatment of periodic alternating nystagmus. Ann Neurol. 1980;8:609–611.
67. Fukazawa T, Tashiro K, Hamada T, Kase M. Multisystem degeneration: drugs and square wave jerks. Neurology. 1986;36:1230–1233.
68. Sharpe JA, Goldberg HJ, Lo AW, Herishanu YO. Visualvestibular interaction in multiple sclerosis. Neurology. 1981;31:427–433.
69. Chambers BR, Gresty MA. The relationship between disordered pursuit and vestibulo-ocular reflex suppression. J Neurol Neurosurg Psychiatry. 1983;46:61–66.